Glanzmann thrombasthenia (GT) is one of many conditions that can cause purpura in childhood, but it is rarely evaluated by dermatologists in its early phases. We describe the case of an infant in whom bruising and facial eczema were key to the early diagnosis of this platelet disorder.1
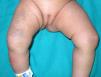
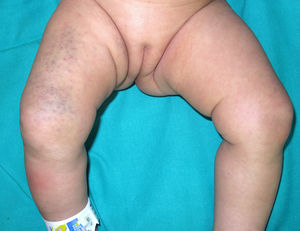
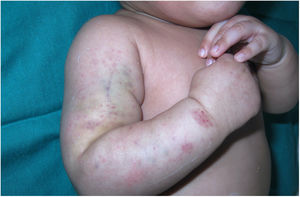
A 2-month-old atopic girl, the first-born daughter of healthy consanguineous parents of Moroccan origin, was evaluated at our dermatology department for purple macules on the right thigh of 2 days’ duration (Fig. 1). The parents attributed the spots to minor trauma and mentioned similar lesions at birth. Physical examination showed eczema on the face and in the retroauricular region. Soft tissue ultrasound of the leg ruled out a venous malformation. Platelet count, international normalized ratio, prothrombin time, and a peripheral blood smear were all normal.
Histologic examination showed extravasation of red blood cells. Tumors and vascular malformations were ruled out.
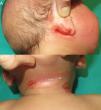
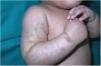
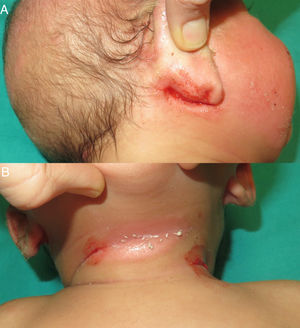
The infant was brought in again a month later for evaluation of bleeding within the eczema patches (Fig. 2) and bruising in pressure-bearing areas (Fig. 3). Additional blood tests were ordered. Abnormal platelet aggregation was observed for all agonists except ristocetin, and the cells were negative for CD61 (integrin iiib). Molecular analysis detected previously undescribed mutations in the ITGB3 gene. A diagnosis of GT was established and genetic counseling was provided to the parents, who have another daughter with the same condition and an asymptomatic son awaiting genetic tests.
GT is an inherited autosomal recessive disorder that presents with bleeding manifestations secondary to impaired platelet aggregation due to quantitative or qualitative defects in the fibrinogen receptor αIIbα3 integrin (glycoprotein IIb/IIIa) in the platelet membrane. The estimated prevalence of GT is 1 case per 1000000 inhabitants, but rates are much higher in areas where consanguinity is common.2–5
Because platelet count and size and coagulation times are normal, additional tests are needed. Platelet aggregation tests show abnormal responses to all agonists except ristocetin. A PFA-100 value of more than 300 in platelet function analyzer tests has a high negative predictive value for GT. Mutations producing very similar phenotypes have been identified in the ITGA2B and ITGB3 genes, both located on chromosome 17.4,6 GT can present with milder symptoms and may not appear until adolescence or adulthood in cases of low penetrance.2,5,7
GT usually manifests before the second year of life, but a diagnosis is not normally made until around 7 years of age. The most common presenting sign in children is bruising caused by minor trauma, but other signs are epistaxis, purpura, and bleeding.5
The differential diagnosis for skin lesions should include thrombocytopenic purpura (triggered by drugs, immune system disorders, infections, or hematologic malignancies), diseases secondary to qualitative platelet disorders (von Willebrand disease, Bernard-Soulier or Chédiak-Higashi), and vasculitides that can present with purpura (e.g., IgA purpura). Trauma and potential abuse should also be contemplated. Given the tendency of eczema patches to bleed, Wiskott-Aldrich syndrome, an immunodeficiency disorder that causes thrombocytopenia and skin lesions similar to those seen in atopic dermatitis, should also be ruled out.1,8,9
Patients with GT normally only require prophylactic treatment before surgical procedures with a high bleeding risk. Options include platelet transfusions, antifibrinolytic agents, and local hemostatic agents. Allogeneic hematopoietic stem cell transplantation may be needed in some cases. Platelet transfusion can lose its effect as it frequently induces antibodies against αIIbβ3 integrin and/or HLA. Recombinant activated factor vii has been proposed for the treatment and prevention of bleeding in GT as well as for the prevention of alloimmunization and treatment of platelet refractoriness. Education in dental hygiene and bleeding management is important. Women with GT should be provided with genetic counseling and given the option of prenatal diagnosis when both parents are carriers. They should also be warned about the risk of bleeding in childbirth.3,10
There is little mention of GT in the dermatology literature, probably due to the nonspecific nature of its skin manifestations. It is important to be familiar with this condition and to consider it in the differential diagnosis of purpura in pediatric patients, as this is the most common presenting manifestation and could help reach an early diagnosis. Bleeding of eczema patches was key to diagnosis in our patient. This sign might be a useful marker for identifying patients with an underlying platelet disorder.
Conflicts of InterestThe authors declare that they have no conflicts of interest.