Postirradiation morphea is an uncommon entity that has been mostly described in women with breast cancer. The increasing use of radiotherapy to treat breast cancer and the clinical similarities between morphea and other conditions, such as radiodermatitis, postirradiation fibrosis, and tumor recurrence, highlights the need for dermatologists to be familiar with this entity. We present a series of 6 women with a mean age of 64.2 years and a mean latency of 9.5 years between radiotherapy for breast cancer and onset of morphea. Four of the patients had a history of autoimmune disease: rheumatoid arthritis, Sjögren syndrome, vitiligo, and Crohn disease. No specific risk factors for postirradiation morphea have been identified to date, although it would appear that a history of autoimmune disease could be associated with an increased risk of morphea in patients treated with radiation therapy.
La morfea postradioterapia es una entidad poco frecuente descrita mayoritariamente en mujeres con cáncer de mama. El uso creciente de la radioterapia en el tratamiento del cáncer de mama, así como la similitud clínica de la morfea con otros cuadros tales como la radiodermitis, la fibrosis postradiación o la recidiva tumoral, obligan al dermatólogo al conocimiento de esta entidad. Presentamos una serie de 6 mujeres, con una media de edad de 64,2 años y un tiempo medio de latencia entre la radioterapia y la morfea de 9,5 años. Cuatro de las 6pacientes presentaban algún trastorno de base autoinmune (artritis reumatoide, síndrome de Sjögren, vitíligo y enfermedad de Crohn) entre sus antecedentes. Hasta el momento actual no se han encontrado factores de riesgo que predispongan al desarrollo de morfea tras el tratamiento con radioterapia, si bien parece que presentar algún trastorno de base autoinmune puede condicionar la aparición de morfea en pacientes tratados con radioterapia.
Postirradiation morphea, or radio-induced morphea, is a rare complication of radiotherapy first described in 1989. Around 70 cases have been reported to date, mostly in women with breast cancer treated with radiotherapy.1
Postirradiation morphea initially presents as an erythematous, edematous plaque in the irradiated area and sometimes even in adjacent areas. The subsequent stage is characterized by a fibrotic reaction with induration and retraction of the affected skin, which is characterized by a whitish central area and violaceous edges. The center of the affected area shows a loss of adnexal structures.2
The estimated incidence of postirradiation morphea is 2 cases per 1000 breast cancer patients treated with radiotherapy.3 This figure contrasts with that of 2.7 cases of morphea per 100000 persons in the general population.4
In view of the growing use of radiotherapy in patients who undergo conservative surgery for breast cancer2 and the clinical similarities between postirradiation morphea and conditions such as radiodermatitis, fibrosis, postirradiation panniculitis, and tumor recurrence, familiarity with this rare, but probably underdiagnosed,5 condition is essential.
We describe a series of 6 women with breast cancer who developed postirradiation morphea. Four of them had a history of autoimmune disease.
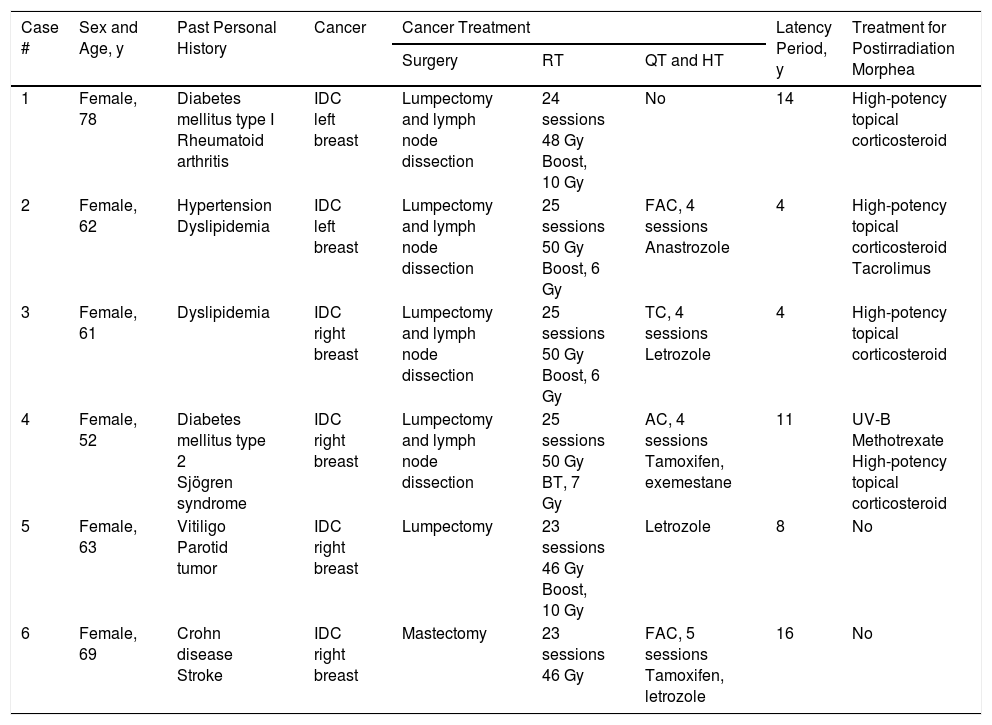
Case DescriptionsWe reviewed all cases of postirradiation morphea referred to the dermatology department at our hospital between 2013 and 2016. We identified 6 cases, all corresponding to women with breast cancer diagnosed and treated between 2000 and 2015. Details of age, medical history, type of cancer and treatment received, and time from administration of radiotherapy to onset of morphea are given in Table 1. The mean age was 64.2 years and the mean latency between radiotherapy and morphea onset was 9.5 years. Four patients had a past personal history of autoimmune disease.
Past Personal History and Time From Radiation Therapy to Onset of Postirradiation Morphea (Latency) in the Present Series.
| Case # | Sex and Age, y | Past Personal History | Cancer | Cancer Treatment | Latency Period, y | Treatment for Postirradiation Morphea | ||
|---|---|---|---|---|---|---|---|---|
| Surgery | RT | QT and HT | ||||||
| 1 | Female, 78 | Diabetes mellitus type I Rheumatoid arthritis | IDC left breast | Lumpectomy and lymph node dissection | 24 sessions 48 Gy Boost, 10 Gy | No | 14 | High-potency topical corticosteroid |
| 2 | Female, 62 | Hypertension Dyslipidemia | IDC left breast | Lumpectomy and lymph node dissection | 25 sessions 50 Gy Boost, 6 Gy | FAC, 4 sessions Anastrozole | 4 | High-potency topical corticosteroid Tacrolimus |
| 3 | Female, 61 | Dyslipidemia | IDC right breast | Lumpectomy and lymph node dissection | 25 sessions 50 Gy Boost, 6 Gy | TC, 4 sessions Letrozole | 4 | High-potency topical corticosteroid |
| 4 | Female, 52 | Diabetes mellitus type 2 Sjögren syndrome | IDC right breast | Lumpectomy and lymph node dissection | 25 sessions 50 Gy BT, 7 Gy | AC, 4 sessions Tamoxifen, exemestane | 11 | UV-B Methotrexate High-potency topical corticosteroid |
| 5 | Female, 63 | Vitiligo Parotid tumor | IDC right breast | Lumpectomy | 23 sessions 46 Gy Boost, 10 Gy | Letrozole | 8 | No |
| 6 | Female, 69 | Crohn disease Stroke | IDC right breast | Mastectomy | 23 sessions 46 Gy | FAC, 5 sessions Tamoxifen, letrozole | 16 | No |
Abbreviations: AC, adriamycin+cyclophosphamide; BT, brachytherapy; FAC, fluorouracil+AC; Gy, gray; HT, hormone therapy; IDC, invasive ductal carcinoma; QT, chemotherapy; RT, radiotherapy; TC, docetaxel+cyclophosphamide; UV, ultraviolet.
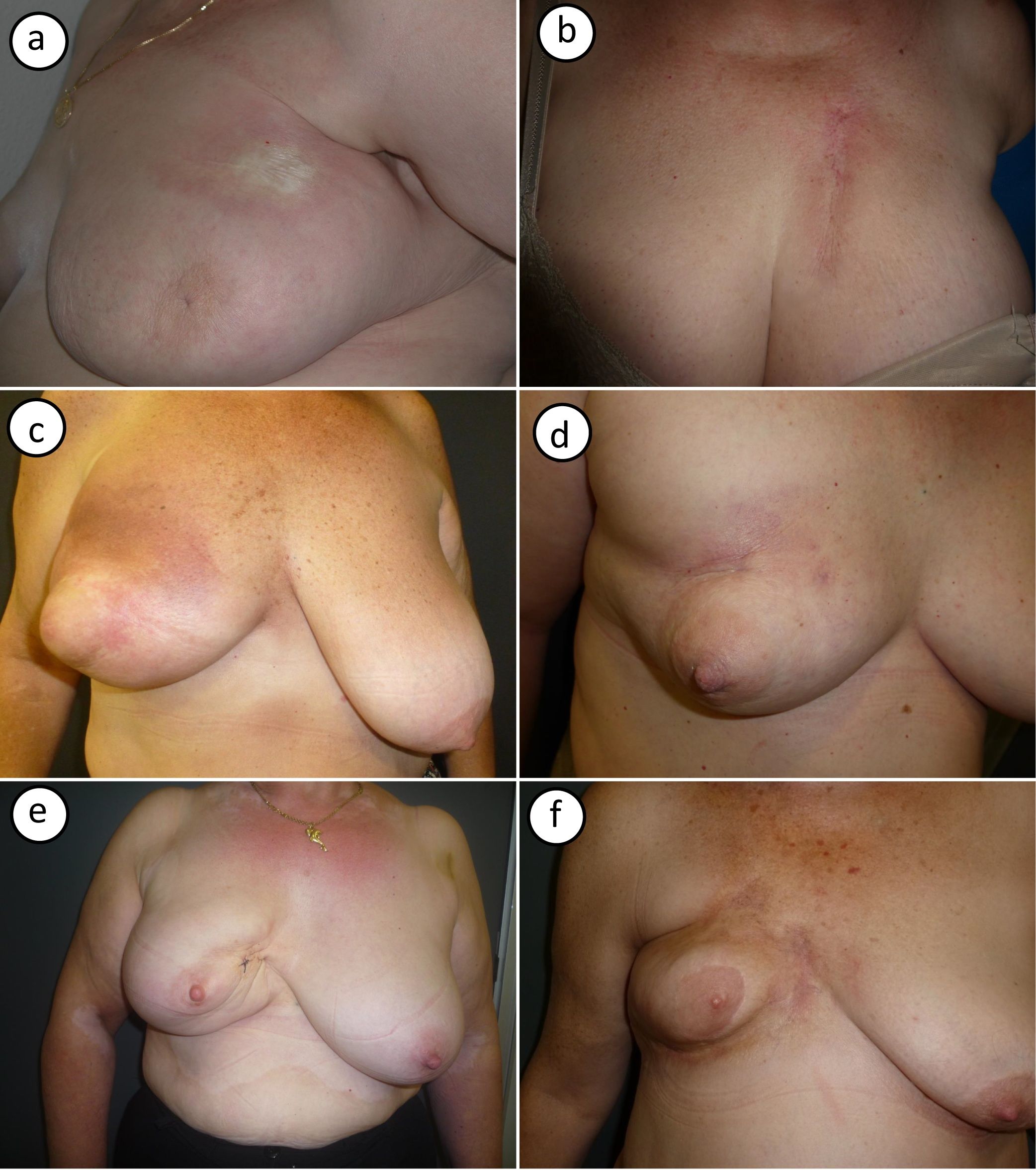
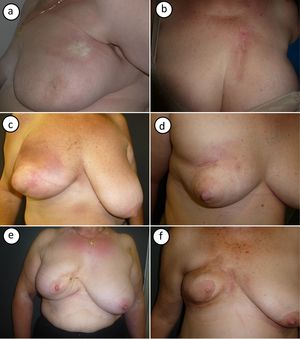
Photographs of the 6 cases are shown in Figure 1. Typical clinical findings for postirradiation morphea were observed and included ivory-colored plaques with a violaceous border (Fig. 1A) and an indurated plaque with signs of radiodermatitis on the surface (Fig. 1B). Two patients showed signs of initial inflammation, with erythema and generalized breast induration (Fig. 1C,D). Later-onset forms were characterized by breast retraction and postinflammatory hyperpigmentation (Fig. 1E,F).
Photographs showing the cases of postirradiation morphea in this series. A (case #1), Morphea on the upper part of the breast, near the armpit. Large, round indurated plaque with a smooth surface, an ivory color, and a violaceous border. B (case #2), Morphea on the upper part of the breast. Highly indurated plaque on an area of radiodermatitis. C, D (cases #3, #4), Inflammatory-stage morphea affecting the entire breast. Inflammation with erythema and generalized breast induration, with sparing of the nipple area. E, F (#cases 5,6), Findings compatible with more residual disease. Pronounced breast retraction and postinflammatory hyperpigmentation.
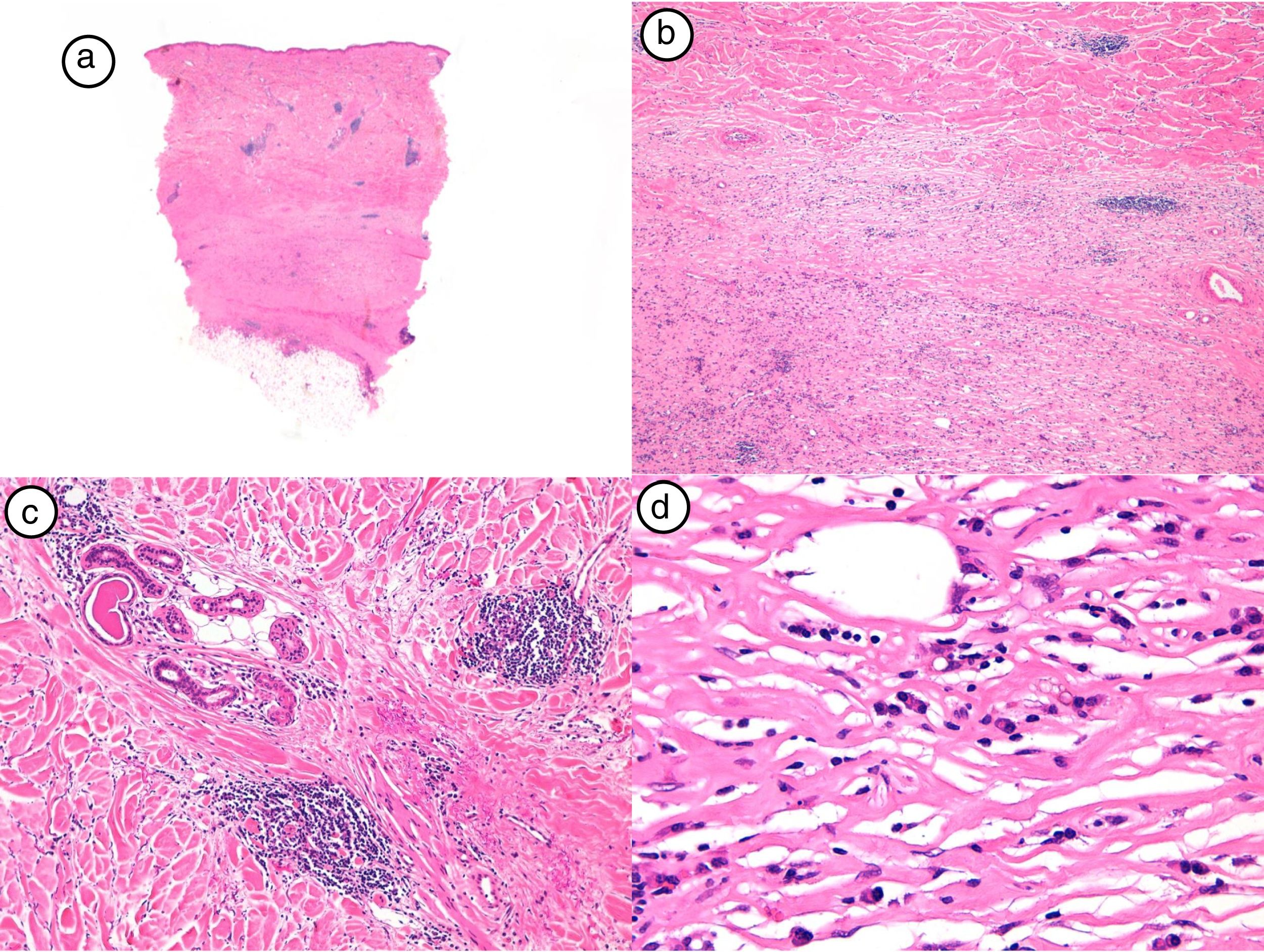
The diagnosis of postirradiation morphea was confirmed by skin biopsy and histologic examination, which showed a periadnexal and perivascular, predominantly lymphocytic, inflammatory infiltrate in the most inflamed lesions in addition to dermal thickening caused by broad collagen bundles. A loss of adnexal structures was observed in the more advanced lesions. The epidermis was spared or just slightly thinned. The characteristic histologic findings of early-stage (inflammatory) postirradiation morphea are shown in Figure 2.
Histologic findings corresponding to case #4. A, Panoramic view with very thickened dermis and little subcutaneous tissue (hematoxylin-eosin [H-E], original magnification ×4). B, Detail of collagen bundle thickening (H-E, original magnification ×40). C, Detail of periadnexal inflammatory infiltrate (H-E, original magnification ×40). D, Plasma cells and lymphocytes forming most of the inflammatory infiltrate (H-E, original magnification ×40).
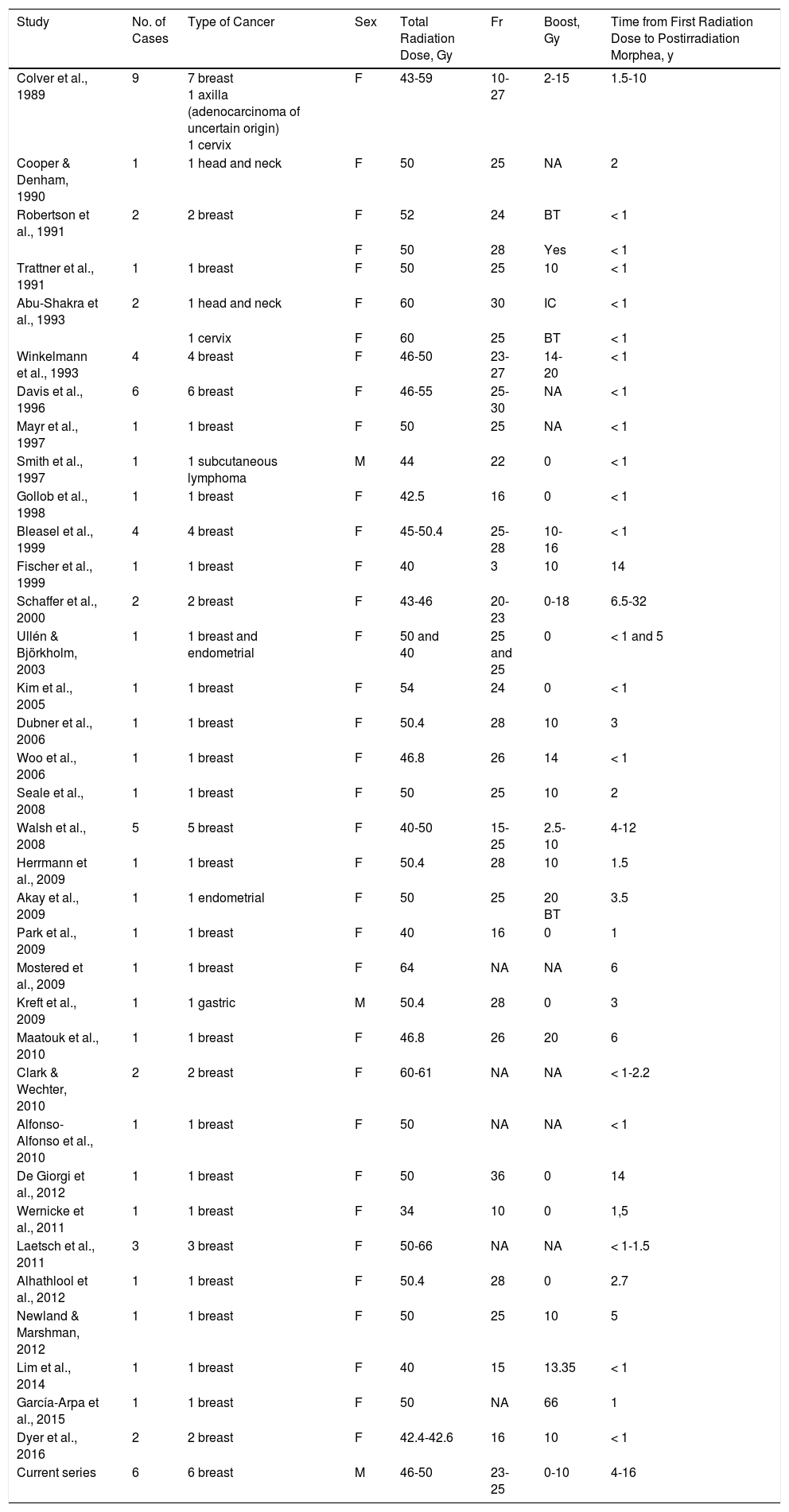
Postirradiation morphea was first described by Colver et al.6 in 1989 in a series of 9 women, 7 of whom had received radiotherapy as part of treatment for breast cancer. Isolated cases have been reported since and are listed together with the 6 cases presented in this article in Table 2. Most of the cases have been in breast cancer patients treated with adjuvant radiotherapy.
Cases of Postirradiation Morphea Published Since 1989.
| Study | No. of Cases | Type of Cancer | Sex | Total Radiation Dose, Gy | Fr | Boost, Gy | Time from First Radiation Dose to Postirradiation Morphea, y |
|---|---|---|---|---|---|---|---|
| Colver et al., 1989 | 9 | 7 breast 1 axilla (adenocarcinoma of uncertain origin) 1 cervix | F | 43-59 | 10-27 | 2-15 | 1.5-10 |
| Cooper & Denham, 1990 | 1 | 1 head and neck | F | 50 | 25 | NA | 2 |
| Robertson et al., 1991 | 2 | 2 breast | F | 52 | 24 | BT | < 1 |
| F | 50 | 28 | Yes | < 1 | |||
| Trattner et al., 1991 | 1 | 1 breast | F | 50 | 25 | 10 | < 1 |
| Abu-Shakra et al., 1993 | 2 | 1 head and neck | F | 60 | 30 | IC | < 1 |
| 1 cervix | F | 60 | 25 | BT | < 1 | ||
| Winkelmann et al., 1993 | 4 | 4 breast | F | 46-50 | 23-27 | 14-20 | < 1 |
| Davis et al., 1996 | 6 | 6 breast | F | 46-55 | 25-30 | NA | < 1 |
| Mayr et al., 1997 | 1 | 1 breast | F | 50 | 25 | NA | < 1 |
| Smith et al., 1997 | 1 | 1 subcutaneous lymphoma | M | 44 | 22 | 0 | < 1 |
| Gollob et al., 1998 | 1 | 1 breast | F | 42.5 | 16 | 0 | < 1 |
| Bleasel et al., 1999 | 4 | 4 breast | F | 45-50.4 | 25-28 | 10-16 | < 1 |
| Fischer et al., 1999 | 1 | 1 breast | F | 40 | 3 | 10 | 14 |
| Schaffer et al., 2000 | 2 | 2 breast | F | 43-46 | 20-23 | 0-18 | 6.5-32 |
| Ullén & Björkholm, 2003 | 1 | 1 breast and endometrial | F | 50 and 40 | 25 and 25 | 0 | < 1 and 5 |
| Kim et al., 2005 | 1 | 1 breast | F | 54 | 24 | 0 | < 1 |
| Dubner et al., 2006 | 1 | 1 breast | F | 50.4 | 28 | 10 | 3 |
| Woo et al., 2006 | 1 | 1 breast | F | 46.8 | 26 | 14 | < 1 |
| Seale et al., 2008 | 1 | 1 breast | F | 50 | 25 | 10 | 2 |
| Walsh et al., 2008 | 5 | 5 breast | F | 40-50 | 15-25 | 2.5-10 | 4-12 |
| Herrmann et al., 2009 | 1 | 1 breast | F | 50.4 | 28 | 10 | 1.5 |
| Akay et al., 2009 | 1 | 1 endometrial | F | 50 | 25 | 20 BT | 3.5 |
| Park et al., 2009 | 1 | 1 breast | F | 40 | 16 | 0 | 1 |
| Mostered et al., 2009 | 1 | 1 breast | F | 64 | NA | NA | 6 |
| Kreft et al., 2009 | 1 | 1 gastric | M | 50.4 | 28 | 0 | 3 |
| Maatouk et al., 2010 | 1 | 1 breast | F | 46.8 | 26 | 20 | 6 |
| Clark & Wechter, 2010 | 2 | 2 breast | F | 60-61 | NA | NA | < 1-2.2 |
| Alfonso-Alfonso et al., 2010 | 1 | 1 breast | F | 50 | NA | NA | < 1 |
| De Giorgi et al., 2012 | 1 | 1 breast | F | 50 | 36 | 0 | 14 |
| Wernicke et al., 2011 | 1 | 1 breast | F | 34 | 10 | 0 | 1,5 |
| Laetsch et al., 2011 | 3 | 3 breast | F | 50-66 | NA | NA | < 1-1.5 |
| Alhathlool et al., 2012 | 1 | 1 breast | F | 50.4 | 28 | 0 | 2.7 |
| Newland & Marshman, 2012 | 1 | 1 breast | F | 50 | 25 | 10 | 5 |
| Lim et al., 2014 | 1 | 1 breast | F | 40 | 15 | 13.35 | < 1 |
| García-Arpa et al., 2015 | 1 | 1 breast | F | 50 | NA | 66 | 1 |
| Dyer et al., 2016 | 2 | 2 breast | F | 42.4-42.6 | 16 | 10 | < 1 |
| Current series | 6 | 6 breast | M | 46-50 | 23-25 | 0-10 | 4-16 |
The time between radiotherapy and onset of the morphea ranges from several months to several years, but there have been cases reported up to 32 years later. Most cases in the literature, however, occur within a year of radiotherapy.7 In our study, the mean latency was 9.5 years (range, 4-16 years). The most widely accepted hypothesis to explain the delay in morphea onset is that radiotherapy induces neoantigens that are not noticed by the immune system until something like an infection triggers cross-reactivity, enabling the immune system to recognize the new antigens in the skin.7
Several studies have investigated potential risk factors for postirradiation morphea, but no association has been found for total dose administered, dose fractionation, or total number of sessions.3,8 There also appears to be no connection with age or the occurrence of acute skin reactions during radiotherapy.8,9
In our series, 4 of the 6 patients had a history of autoimmune disease, namely, rheumatoid arthritis, Sjögren syndrome, vitiligo, and Crohn disease. In 2011, Laetsch et al.10 described a patient with a history of alopecia areata and vulval and cutaneous lichen sclerosus et atrophicus who developed a lesion consistent with postirradiation morphea. Laetsch and colleagues were the first to suggest that patients with autoimmune disease may be at greater risk of connective tissue disease (CTD) following radiotherapy.10 Postirradiation morphea has also been reported in patients with a history of morphea and psoriasis11 and CREST syndrome (calcinosis, Raynaud's phenomenon, oesophageal dysmotility, sclerodactyly and telangiectasia).12 Most cases, however, make no mention of personal history.
Patients with CTD appear to have an increased risk of radiotherapy-associated complications. In 1991, Varga et al.13 described 4 patients with systemic sclerosis who developed severe fibrosis in irradiated areas with cutaneous and internal involvement. Three of the patients died of related complications. In another study, Lin et al.14 found that patients with CTD had a greater risk of late toxicity from radiotherapy compared with the general population. The incidence of late toxicity was particularly remarkable in patients with systemic lupus erythematosus (35.3% vs 4.8% for controls) and scleroderma (10% vs 3.9%).14
Radiotherapy can induce or worsen CTD. This treatment has been found to induce sclerosing panniculitis, bullous pemphigoid, pemphigoid vulgaris, dermatomyositis, and systemic lupus erythematous.10 In view of the increased risk of CTD flare-ups and more severe complications associated with radiotherapy, a rheumatologic history should be taken in patients scheduled to undergo this treatment.15
Treatment outcomes in patients with postirradiation morphea vary across the literature. The treatments described include topical, intralesional, and oral corticosteroids, calcineurin inhibitors, phototherapy, and immunosuppressive agents. Outcomes, however, are not always satisfactory, with some patients responding well, and others failing to respond.1
In conclusion, postirradiation morphea is a rare condition but its incidence is likely to rise in coming years. We have presented the second largest series of postirradiation morphea cases to date and would like to draw attention to the high proportion of patients with a past personal history of autoimmune disease. More studies are needed to assess whether autoimmune disease is a risk factor for postirradiation morphea, especially in the setting of breast cancer. The existence of additional, yet-to-be discovered, risk factors also needs to be investigated.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as: Diago A, Llombart B, Requena C, Sanmartín O, Guillén C. Postirradiation Morphea in Patients With Breast Cancer: Possible Association With Other Autoimmune Diseases. Actas Dermosifiliogr. 2019;110:153–159.

![Histologic findings corresponding to case #4. A, Panoramic view with very thickened dermis and little subcutaneous tissue (hematoxylin-eosin [H-E], original magnification ×4). B, Detail of collagen bundle thickening (H-E, original magnification ×40). C, Detail of periadnexal inflammatory infiltrate (H-E, original magnification ×40). D, Plasma cells and lymphocytes forming most of the inflammatory infiltrate (H-E, original magnification ×40). Histologic findings corresponding to case #4. A, Panoramic view with very thickened dermis and little subcutaneous tissue (hematoxylin-eosin [H-E], original magnification ×4). B, Detail of collagen bundle thickening (H-E, original magnification ×40). C, Detail of periadnexal inflammatory infiltrate (H-E, original magnification ×40). D, Plasma cells and lymphocytes forming most of the inflammatory infiltrate (H-E, original magnification ×40).](https://static.elsevier.es/multimedia/15782190/0000011000000002/v1_201903030657/S1578219019300083/v1_201903030657/en/main.assets/thumbnail/gr2.jpeg?xkr=ue/ImdikoIMrsJoerZ+w9/t1/zx4Q/XH5Tma1a/6fSs=)


