Sentinel lymph node (SLN) biopsy is a staging, not a therapeutic, procedure. The benefits of SLN biopsy have been more modest than expected and could be outweighed by the risks, which remain unclear. The aim of this study was to describe complications and sequelae observed in patients with melanoma who underwent routine SLN biopsy at our hospital.
Patients and methodsIn this retrospective cohort study, we performed a chart review of all patients with melanoma who underwent SLN biopsy at our hospital in Vigo, Spain, between January 2011 and July 2017.
ResultsIn the period analyzed, 124 SLN biopsies were performed. Over a mean follow-up of 52.7 months (range, 10.8-88.7 months). A percentage of 37.9 of the patients experienced complications. The complication rate after excluding patients who underwent lymph node dissection was 30.9%. In the full cohort group, there were 14 scar-related complications (11.3%), 13 surgical wound infections (10.5%), 12 lymphedemas (9.7%), 11 seromas (8.9%), 4 hematomas (3.2%), 4 wound dehiscences (3.2%), 2 cases of lymphorrhagia (1.6%), 2 cases of sensitivity alteration (1.6%), and 1 urinary tract infection (0.8%). The most common sequela was lymphedema. Sequelae were on record for 15.3% of patients in the full cohort (7.5% of the patients who did not undergo lymphadenectomy). Smoking was associated with a 33% to 73% increased risk of complications. The main limitation of this study is the risk of information bias due to incomplete follow-up.
ConclusionsSLN biopsy is a melanoma staging procedure that causes complications and sequelae. Recommendations for its use in clinical practice guidelines should be revised and the risks and benefits carefully evaluated in each case. Smokers, in particular, seem to have a high risk of complications. Patients with melanoma could benefit greatly from the development of less invasive staging tools.
La biopsia selectiva de ganglio centinela (BSGC) no tiene utilidad terapéutica y solo se utiliza por su valor pronóstico. Su beneficio ha sido menor del esperado, por lo que los riesgos cobran más valor y no están claramente definidos. Nos hemos propuesto describir las complicaciones y secuelas sufridas por pacientes con melanoma expuestos a la técnica de BSGC en la práctica clínica habitual.
Pacientes y métodosEn este estudio de cohortes retrospectivo unicéntrico hemos recogido los datos de todos los pacientes diagnosticados de melanoma y sometidos a la BSGC en nuestro centro (Vigo) entre enero de 2011 y julio de 2017, revisando sus historias clínicas.
ResultadosSe realizaron 124 BSGC. El tiempo de seguimiento medio fue de 52,7 meses (rango 10,8-88,7 meses). El 37,9% de los pacientes presentaron complicaciones. Excluyendo a aquellos en los que se realizó linfadenectomía, el porcentaje de complicaciones fue del 30,9%. De las complicaciones totales, 14 (11,3%) fueron alteraciones en la cicatrización, 13 (10,5%) infecciones de la herida quirúrgica, 12 (9,7%) linfedemas, 11 (8,9%) seromas, 4 (3,2%) hematomas, 4 (3,2%) heridas dehiscentes, 2 (1,6%) linforragias, 2 (1,6%) alteraciones sensitivas y una (0,8%) infección del tracto urinario. El 15,3% de los pacientes presentaron secuelas, siendo el linfedema la más frecuente. Entre los pacientes sin linfadenectomía completa presentaron secuelas el 7,5%. El tabaquismo se asoció con un aumento en el porcentaje de complicaciones de un 33 a un 73%. La principal limitación de este estudio es que pueda haber un sesgo de información que infravalore los resultados por un seguimiento incompleto de los pacientes.
ConclusionesLa BSGC es una técnica de estadificación del melanoma no exenta de complicaciones y secuelas. La recomendación de su uso rutinario en las guías de práctica clínica debería revisarse, sopesando los riesgos y los beneficios en cada caso. En especial tienen un alto riesgo de presentar complicaciones los pacientes fumadores. El desarrollo de otras herramientas de estadificación menos invasivas puede ser de gran utilidad para los pacientes con melanoma.
Sentinel lymph node (SLN) biopsy is a minimally invasive procedure, but there is disagreement about its potential complications.
The American Society of Clinical Oncology (ASCO) and Society of Surgical Oncology (SSO) clinical practice guideline on the use of SLN biopsy in melanoma states that this technique may be considered for T1b melanomas (Breslow thickness, 0.8-1mm or <0.8mm with ulceration) after thorough discussion of risks and benefits with the patient. The guideline recommends the procedure for T2 and T3 melanomas (Breslow thickness, 1-4mm) and states that it may be recommended for T4 melanomas (Breslow thickness, >4mm), again after weighing up the risks and benefits with the patient. If the biopsy is positive, patients can be treated with complete lymph node dissection (CLND) or careful observation depending on the risk of micrometastatic disease, determined by an evaluation of clinical and pathologic factors.1
The European Society of Medical Oncology (ESMO) clinical practice guidelines recommend SLN biopsy for melanomas with a Breslow thickness of over 1mm or of over 0.75mm if there are additional risk factors, such as ulceration and a mitotic rate of 1 mitosis/mm2 (pT1b).2 In this case, the guidelines state that CLND must be considered if the biopsy is positive.
The above recommendation is made despite a lack of evidence showing the therapeutic value of SLN biopsy, followed or not by CLND, as the procedure has not been found to improve overall survival in patients with melanoma.3 Its usefulness therefore lies in its ability to provide prognostic information.
Although SLN biopsy causes less morbidity than CLND, its complication rates are still considerable. In addition, patients with a positive result were until very recently offered CLND and hence exposed to the risk of further complications.
Several studies in recent years have evaluated the therapeutic and prognostic value of SLN biopsy and its associated complications.4–13 Their results highlight variations in study design and complication rates ranging from 4.6%9 to 33%.6 There is thus no clear evidence on the true incidence of complications and sequelae associated with this technique.
The aim of this study was to describe the incidence of complications and sequelae in patients with melanoma who underwent SLN biopsy.
MethodsStudy Design and ScopeRetrospective cohort study of all patients with melanoma in the healthcare district of Vigo (northwest Spain) who underwent SLN biopsy between January 1, 2011 and June 5, 2017. The patients were identified from the pathology laboratory database at our hospital.
We also analyzed the subgroup of patients who underwent CLND after a positive SLN biopsy, as we consider that complications caused by CLND are indirectly linked to the indication of SLN biopsy, as none of the patients would have undergone CLND if the biopsy had not been performed.
We recorded the following information for all patients: demographic variables, tumor characteristics, SLN biopsy characteristics, including assessment of whether the biopsy was performed as indicated by current guidelines (using the ASCO/SSO guideline1 as a reference), biopsy results, performance of CLND in a second procedure, and complications and sequelae reported in the patient's medical record. Sequelae were defined as adverse effects lasting over 1 year. The definitions of complications and sequelae are described in the supplementary material (see Appendix B supplementary material). Severity was graded using the classification system described by Dindo et al.14 in 2004, which is based on the type of intervention needed to treat the complication and the associated risk to the patient's life (see Appendix B supplementary material). Lymphedema severity was assessed using the International Society of Lymphology classification system.15
Data were obtained from electronic medical records and recorded anonymously using EpiData.
The statistical analyses were performed in Stata 14. We analyzed the risk of different complications and sequelae associated with SLN biopsy. Descriptive statistics for the population are presented as mean (SD) in the case of quantitative variables and percentages in the case of qualitative variables.
Qualitative variables were compared using the Fisher exact test. Logistic regression was used to explore significant associations (P≤.05) between variables and the occurrence of complications.
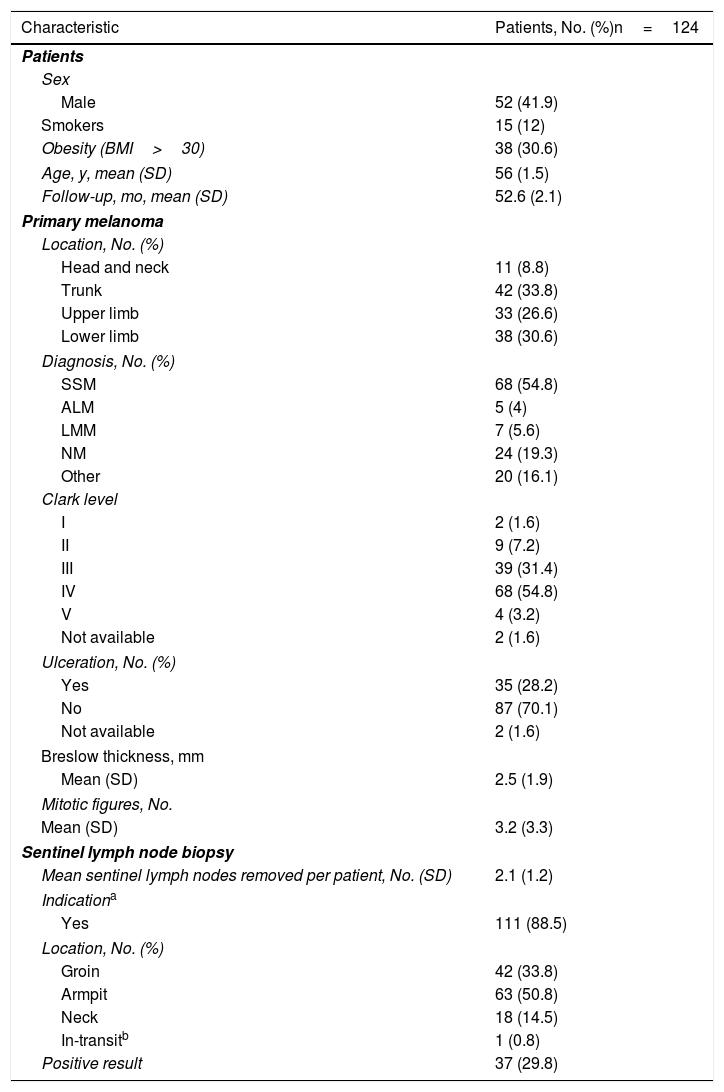
ResultsPopulation and Follow-upWe identified 124 patients who underwent SLN biopsy during the study period. Their characteristics are summarized in Table 1.
Demographic Variables, Clinical and Pathologic Characteristics of Primary Melanomas, and Details of Sentinel Lymph Node Biopsy.
| Characteristic | Patients, No. (%)n=124 |
|---|---|
| Patients | |
| Sex | |
| Male | 52 (41.9) |
| Smokers | 15 (12) |
| Obesity (BMI>30) | 38 (30.6) |
| Age, y, mean (SD) | 56 (1.5) |
| Follow-up, mo, mean (SD) | 52.6 (2.1) |
| Primary melanoma | |
| Location, No. (%) | |
| Head and neck | 11 (8.8) |
| Trunk | 42 (33.8) |
| Upper limb | 33 (26.6) |
| Lower limb | 38 (30.6) |
| Diagnosis, No. (%) | |
| SSM | 68 (54.8) |
| ALM | 5 (4) |
| LMM | 7 (5.6) |
| NM | 24 (19.3) |
| Other | 20 (16.1) |
| Clark level | |
| I | 2 (1.6) |
| II | 9 (7.2) |
| III | 39 (31.4) |
| IV | 68 (54.8) |
| V | 4 (3.2) |
| Not available | 2 (1.6) |
| Ulceration, No. (%) | |
| Yes | 35 (28.2) |
| No | 87 (70.1) |
| Not available | 2 (1.6) |
| Breslow thickness, mm | |
| Mean (SD) | 2.5 (1.9) |
| Mitotic figures, No. | |
| Mean (SD) | 3.2 (3.3) |
| Sentinel lymph node biopsy | |
| Mean sentinel lymph nodes removed per patient, No. (SD) | 2.1 (1.2) |
| Indicationa | |
| Yes | 111 (88.5) |
| Location, No. (%) | |
| Groin | 42 (33.8) |
| Armpit | 63 (50.8) |
| Neck | 18 (14.5) |
| In-transitb | 1 (0.8) |
| Positive result | 37 (29.8) |
Abbreviations: ALM, acral lentiginous melanoma; BMI, body mass index; LLM, lentigo maligna melanoma; NM, nodular melanoma; SSM, superficial spreading melanoma.
SLN biopsy was performed according to the indications in the ASCO-SSO clinical practice guideline in 111 (89.5%) of the patients. In the remaining 13 cases (10.4%), the indication was unclear. Local excision with wide margins was performed at the same time as the biopsy in 95.1% of cases (118/124).
In total, 24.1% of patients in the overall group (30/124) and 81% of those with a positive SLN biopsy (30/37) underwent CLND.
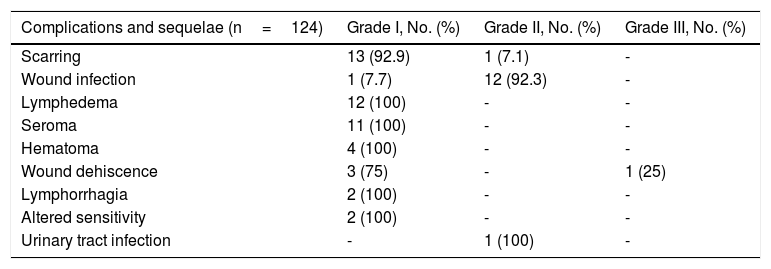
ComplicationsComplications were reported for 37.9% of patients (47/124) (95% CI, 29.7%-46.9%), and several patients had more than 1 complication. The type and severity are summarized in Table 2.
Severity of Complications and Sequelae.
| Complications and sequelae (n=124) | Grade I, No. (%) | Grade II, No. (%) | Grade III, No. (%) |
|---|---|---|---|
| Scarring | 13 (92.9) | 1 (7.1) | - |
| Wound infection | 1 (7.7) | 12 (92.3) | - |
| Lymphedema | 12 (100) | - | - |
| Seroma | 11 (100) | - | - |
| Hematoma | 4 (100) | - | - |
| Wound dehiscence | 3 (75) | - | 1 (25) |
| Lymphorrhagia | 2 (100) | - | - |
| Altered sensitivity | 2 (100) | - | - |
| Urinary tract infection | - | 1 (100) | - |
No life-threatening (grade iv) or fatal (grade v) complications were observed. Most of the complications were grade i or ii and there was 1 grade iii complication.
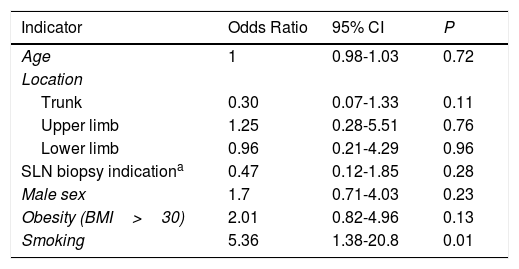
In the multivariate logistic regression analysis, no differences were observed for occurrence of complications according to age, sex, obesity, SLN biopsy indication according to the ASCO/SSO guideline, or melanoma site (Table 3). Smoking, however, was associated with an increased odds of experiencing a complication (odds ratio [OR], 5.4; 95% CI, 1.4-20.8; P=.015); 33.0% (95% CI, 24.8%-42.5%) of nonsmokers (36/109) developed complications compared with 73.3% (95% CI, 45.4%-90.1%) of smokers (11/15) (Fisher exact text, P=.004).
Multivariate Logistic Regression Analysis of the Effect of Different Variables on the Risk of Complications Expressed by Odds Ratios.
| Indicator | Odds Ratio | 95% CI | P |
|---|---|---|---|
| Age | 1 | 0.98-1.03 | 0.72 |
| Location | |||
| Trunk | 0.30 | 0.07-1.33 | 0.11 |
| Upper limb | 1.25 | 0.28-5.51 | 0.76 |
| Lower limb | 0.96 | 0.21-4.29 | 0.96 |
| SLN biopsy indicationa | 0.47 | 0.12-1.85 | 0.28 |
| Male sex | 1.7 | 0.71-4.03 | 0.23 |
| Obesity (BMI>30) | 2.01 | 0.82-4.96 | 0.13 |
| Smoking | 5.36 | 1.38-20.8 | 0.01 |
Abbreviations: BMI, body mass index; SLN, sentinel lymph node.
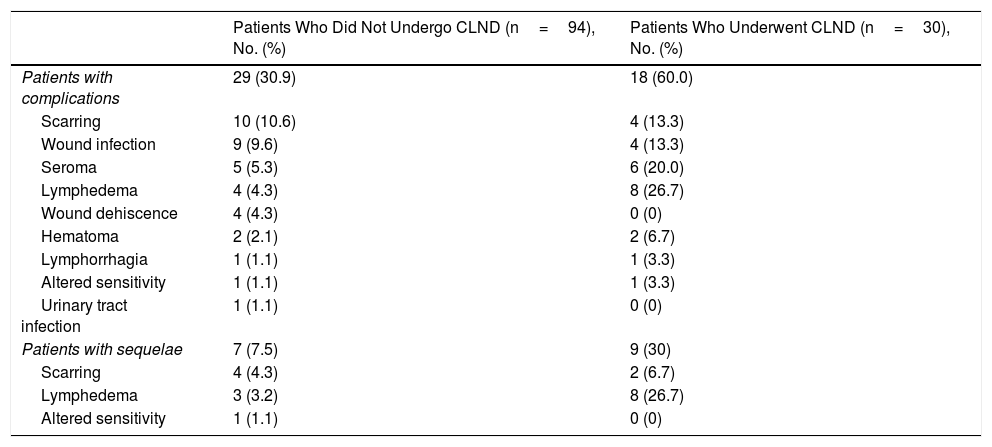
Table 4 shows the incidence of complications according to whether patients underwent SLN biopsy only or SLN biopsy followed by CLND. The rate was 30.9% (95% CI, 22.2%-41%) in patients who underwent SLN biopsy only (29/94) compared with 60% (95% CI, 41.5%-76.1%) in those who underwent SLN biopsy and CLND (18/30) (Fisher exact test, P=.005).
Complications and Sequelae According to Performance of Complete Lymph Node Dissection (CLND).
| Patients Who Did Not Undergo CLND (n=94), No. (%) | Patients Who Underwent CLND (n=30), No. (%) | |
|---|---|---|
| Patients with complications | 29 (30.9) | 18 (60.0) |
| Scarring | 10 (10.6) | 4 (13.3) |
| Wound infection | 9 (9.6) | 4 (13.3) |
| Seroma | 5 (5.3) | 6 (20.0) |
| Lymphedema | 4 (4.3) | 8 (26.7) |
| Wound dehiscence | 4 (4.3) | 0 (0) |
| Hematoma | 2 (2.1) | 2 (6.7) |
| Lymphorrhagia | 1 (1.1) | 1 (3.3) |
| Altered sensitivity | 1 (1.1) | 1 (3.3) |
| Urinary tract infection | 1 (1.1) | 0 (0) |
| Patients with sequelae | 7 (7.5) | 9 (30) |
| Scarring | 4 (4.3) | 2 (6.7) |
| Lymphedema | 3 (3.2) | 8 (26.7) |
| Altered sensitivity | 1 (1.1) | 0 (0) |
No differences were observed between patients in whom SLN biopsy was performed according to guideline indications (38.5% [5/13]; 95% CI, 16.2%-66.9%) and patients for whom the indication was not clear (37.8% [42/111]; 95% CI, 29.2%-47.3%) (Fisher exact test, P=1).
There were also no significant difference between patients with a negative and a positive result (32.2% [28/87]; 95% CI, 23.1%-42.8% vs. 51.4% [19/37]; 95% CI, 35.3%-67.1%) (Fisher exact test, P=.068).
The SLN was identified in all cases.
SequelaeSequelae were recorded for 15.3% of patients (16/124), and the most common sequela was lymphedema (8.9%). Two patients had 2 sequelae: lymphedema and scarring in one case and altered sensitivity and scarring in the other. The incidence of the different types of sequelae is shown in Table 4.
Lymphedema affected the upper arm in 7 patients (63.6%) and the lower arm in 4 (36.4%). Two cases (18.2%) were graded as stage 0, mild severity; 5 (45.4%) as stage I, mild severity; 2 as stage I, moderate severity; and 2 as stage II, moderate severity. Three of the 11 patients who developed lymphedema had not undergone CLND. The association between persistent lymphedema and CLND was not significant (OR, 2.7; 95% CI, 0.7-10.1), although the lack of association could be due to the small number of cases.
One patient developed altered sensitivity in the form of paraesthesia affecting the outer ear and left neck region.
In the subgroup of patients with scar-related complications, 2 patients developed keloid scars that persisted despite treatment and another developed chronic scar tissue pain in the armpit area; 3 patients had persistent limited mobility of the upper arm, 2 had chronic upper arm pain, and 1 had neck rigidity.
Sequelae were less common in patients who did not undergo CLND (7.4% [7/94]; 95% CI 3.6%-15%) than in those who did (30% [9/30], 95% CI, 16.1%-48.9%) (Fisher exact test, P=.003).
No significant differences were observed for the incidence of sequelae in patients who underwent SLN biopsy according to guideline recommendations (15.4% [2/13]; 95% CI, 3.6%-47%) and those who did not (12.6% [14/111]; 95% CI, 7.6%-20.3%) (Fisher exact test, P=.675).
Sequelae were more common in patients with a positive SLN biopsy (24.3% [9/37]; 95% CI, 13%-40.9% vs 8% [7/87]; 95% CI, 3.8%-16.1% for those with a negative biopsy) (Fisher exact test, P=.019).
DiscussionOur study shows that complications and sequelae are common in patients who undergo SLN biopsy.
Comparisons with other studies that have analyzed complications associated with SLN biopsy are difficult due to large differences. Most studies do not provide clear definitions of the complications analyzed, while others describe short-term or long-term complications or complications that require repeat surgery.
This heterogeneity was demonstrated in a meta-analysis of 21 studies by Moody et al,16 who reported an overall complication rate of 11.3% (95% CI, 8.1%-15.0%). The specific rates were 2.9% (95% CI, 1.5%-4.6%) for infections, 5.1% (95% CI, 2.5%-8.6%) for seroma, 0.5% (95% CI, 0.3%-0.9%) for hematoma, 1.3% (95% CI, 0.5%-2.6%) for lymphedema, and 0.3% (95% CI, 0.1%-0.6%) for nerve damage. No significant differences according to SLN biopsy site or geographic location (continent) were observed. The overall complication rate in our series was higher, at 37.9%, but this is partly because we included patients who underwent CLND after SLN biopsy. We believe that this design is important for studying overall SLN biopsy complications, as patients referred for SLN biopsy are likely to undergo CLND if the biopsy result is positive. Findings that CLND does not improve survival in micrometastatic SLN disease suggest that this procedure should no longer be indicated in this setting, but these findings emerged after our study started. Indeed a high proportion of patients with a positive SLN biopsy in our series did not undergo CLND. To enable comparison with future studies we have separated our findings for complications and sequelae according to patients who underwent SLN biopsy only and those who underwent SLN biopsy plus CLND (Table 4).
In the Multicenter Selective Lymphadenectomy Trial 1 (MSLT-1), Morton et al.17 described a complication rate of 10.1% for patients who underwent SLN biopsy only and 37.1% for those who underwent SLN biopsy followed by CLND. These lower rates could be due to the fact that only regional complications that occurred within 30 days of the procedure were considered. The complication rate associated with SLN biopsy rose to 12.6% when systemic complications (leg edema, thrombophlebitis, and urinary, pulmonary and other complications) were also considered. The article does not describe complications or sequelae that occurred after this initial period of 30 days.
In MSLT-2, Faries et al.18 only reported lymphedema rates, which were 6.3% in patients who underwent SLN biopsy and 24.1% in those who underwent SLN biopsy and CLND. In this case, the follow-up time was 3 years and the rates are similar to those observed in our study.
The authors of the DeCOG-SLT trial (CLND versus no dissection in patients with sentinel lymph biopsy–positive melanoma) only reported adverse events for patients who underwent CLND. These events were analyzed immediately after surgery and at 3 and 6 months. Follow-up was longer for moderate and severe complications. The complication rate was 24% (58/240); 6% (15/240) of the complications were moderate, while 8% (19/240) were severe. Moderate and severe complications included lymphedema, lymph fistula, seroma, infection, and delayed wound healing. Lymphedema occurred in 20 patients and persisted in 13 of these (5.4%).19 Rates for both lymphedema and persistent lymphedema in our series were more common, possibly because of the longer follow-up period.
It is difficult to compare complication rates for patients who underwent SLN biopsy only (30.9% in our series) with other studies, as most do not consider scarring complications, such as keloid or fibrous scars, or only report on complications in the immediate postoperative period. In one study that did include scarring, Hettiaratchy et al.6 reported a complication rate of 33%, which is similar to ours.
The only factor significantly associated with an increased likelihood of complications in our series was smoking, as 73.3% of smokers developed complications, compared with just 33.0% of nonsmokers. This result coincides with reports by Ascha et al.,12 who in their study of risk factors for lymphatic surgeries for melanoma found smoking to be the only significant predictor of complications associated with SLN biopsy, regardless of biopsy site (odds ratio, 1.8; 95% CI, 1.0-2.9; P=0.038).
Our study has some limitations, including the risk of information bias that might have affected the accuracy of our results. Patients, for example, may have been treated for complications at another center (although the medical records analyzed cover primary care) or may not have sought care. Likewise, complications may not have been accurately recorded. However, if this bias did exist, the true complication and sequela rates would be even higher. In our opinion, information bias is unlikely as similar rates have been reported in studies with a similar design.6
Another potential limitation is that diagnoses were based on data from medical records, which are less reliable than prospectively collected data based on pre-established definitions. Nevertheless, we do not believe that our retrospective design resulted in an overestimation of complications or sequelae.
Another consideration is that our results are from a single hospital and may not be representative. We did, however, analyze data for all consecutive patients in the database. At our hospital, SLN biopsy is performed by surgeons with extensive experience and we believe that the identification of the SLN in all cases attests to adequate performance of the procedure.
We used the ASCO/SSO clinical practice guideline to determine whether or not the SLN biopsies in our series were performed according to current indications, and while the indication rates may have varied slightly had we used another guideline, we do not believe that this would have altered our results.
The morbidity rates associated with SLN biopsy in our cohort and in previous studies have important implications for the routine use of this procedure as a staging tool. The use of any invasive technique should only be contemplated when the benefits for the patient outweigh the risks. Clinical trials to date have not demonstrated an overall survival benefit for SLN biopsy alone or for biopsy followed by CLND. Therefore, as this procedure provides purely prognostic information, the risk of complications and sequelae may be unacceptable. Techniques and methods with a lower risk, such as analysis of clinical data, imaging studies, and proteomic or genomic studies, could offer similarly useful information.
The indication for CLND in patients with micrometastatic disease was recently called into question. A change in practice in this regard would lead to a reduction in associated morbidity, but according to our findings, SLN biopsy is still associated with complication and sequela rates of 30.9% and 7.5% and rates this high may not be justifiable for a procedure that provides prognostic information only, even if the complications are of moderate severity.
The increased risk of complications in smokers is also noteworthy and suggests that the indications for SLN biopsy in this subgroup of patients should be even more restrictive.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
We thank the pathology department for giving us access to its database, which contains information on all SLN biopsies performed in the healthcare district of Vigo as well as melanoma pathology reports.
Please cite this article as: Espinosa-Pereiro CE, Zulaica Gárate A, García-Doval I. Complicaciones y secuelas de la técnica de biopsia selectiva del ganglio centinela para el melanoma en una cohorte retrospectiva. Actas Dermosifiliogr. 2019;110:482–489.