Facial discoid dermatosis (FDD) is a recently described condition1 that differs from other facial dermatoses in its clinical appearance and histopathologic findings—which mimic those of pityriasis rubra pilaris (PRP)—as well as in its failure to respond to treatment. We describe a characteristic case of FDD.
A 61-year-old man with no remarkable past medical history presented with 3 intensely pruriginous lesions that had appeared 2 months before. He had tried antihistamines, corticosteroids, antibiotics, and topical antifungal creams without seeing improvement.
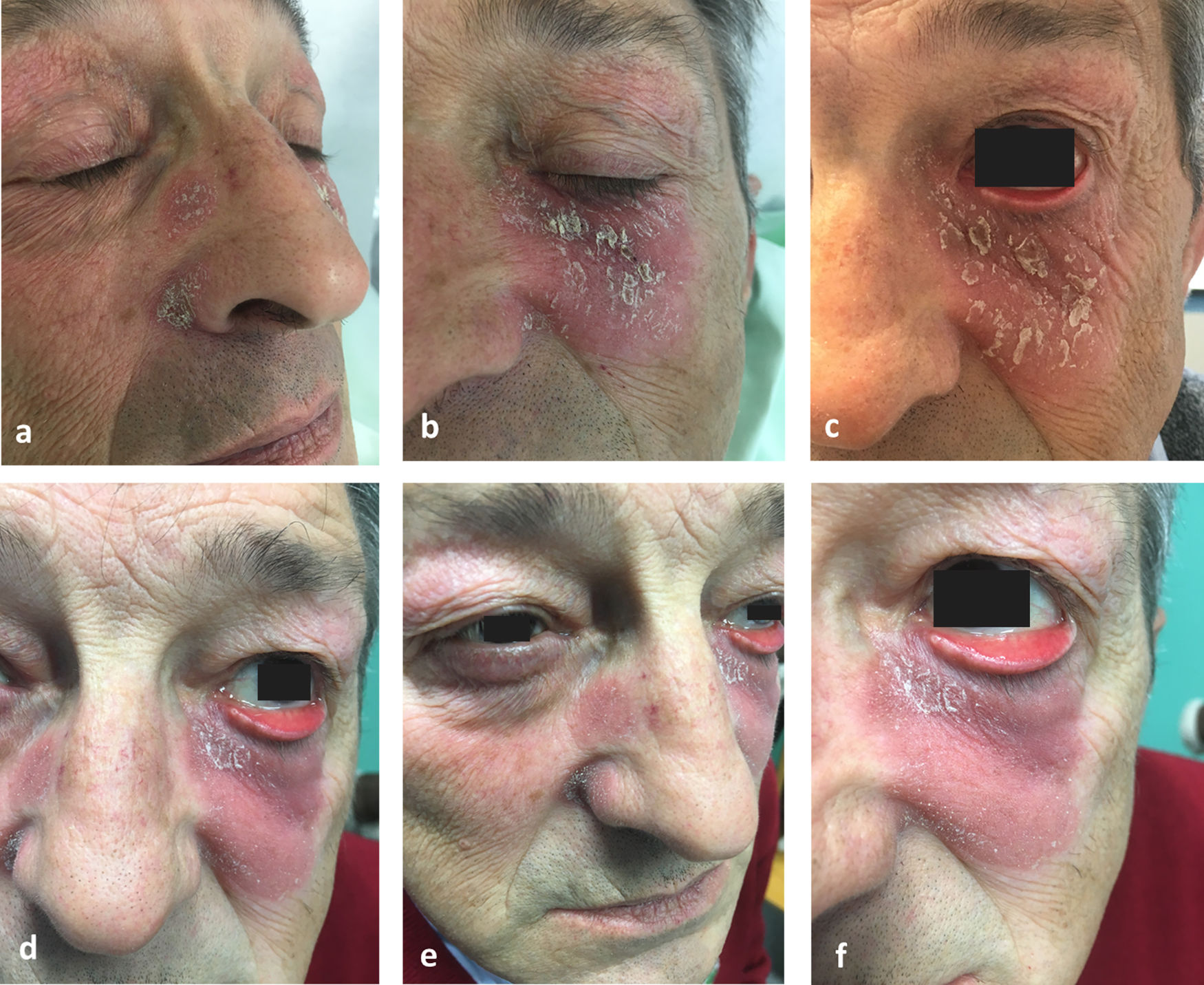
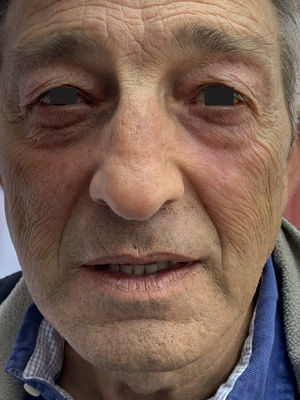
Physical examination showed a salmon-colored erythematous plaque with thick yellowish, adherent scaling. The plaque in the left malar region measured approximately 3cm in diameter and affected the lower eyelid. Two other similar but smaller plaques were observed. They were circular and on the right side of the nose. Involvement of the eyelid progressed and eventually caused marked ectropion (Fig. 1). Two biopsies were performed for histopathology (Fig. 2a and b). The results of laboratory analyses (a full blood work-up including biochemistry, immunoglobulins, and antibodies) were normal or negative, as were patch tests. Successive treatments with clobetasol, prednisone, hydroxychloroquine, and methotrexate were ineffective. Despite poor control of the skin disease, the consulting ophthalmologists scheduled surgical ectropion repair. However, the procedure was not performed because 2 months after the last treatment prescribed — an ointment containing calcipotriol plus betamethasone to be applied at night — all symptoms resolved (Fig. 3). At the time of writing a year later, the patient was applying a 0.1% tacrolimus cream twice a week because repeated attempts to stop treatment altogether were followed by relapses.
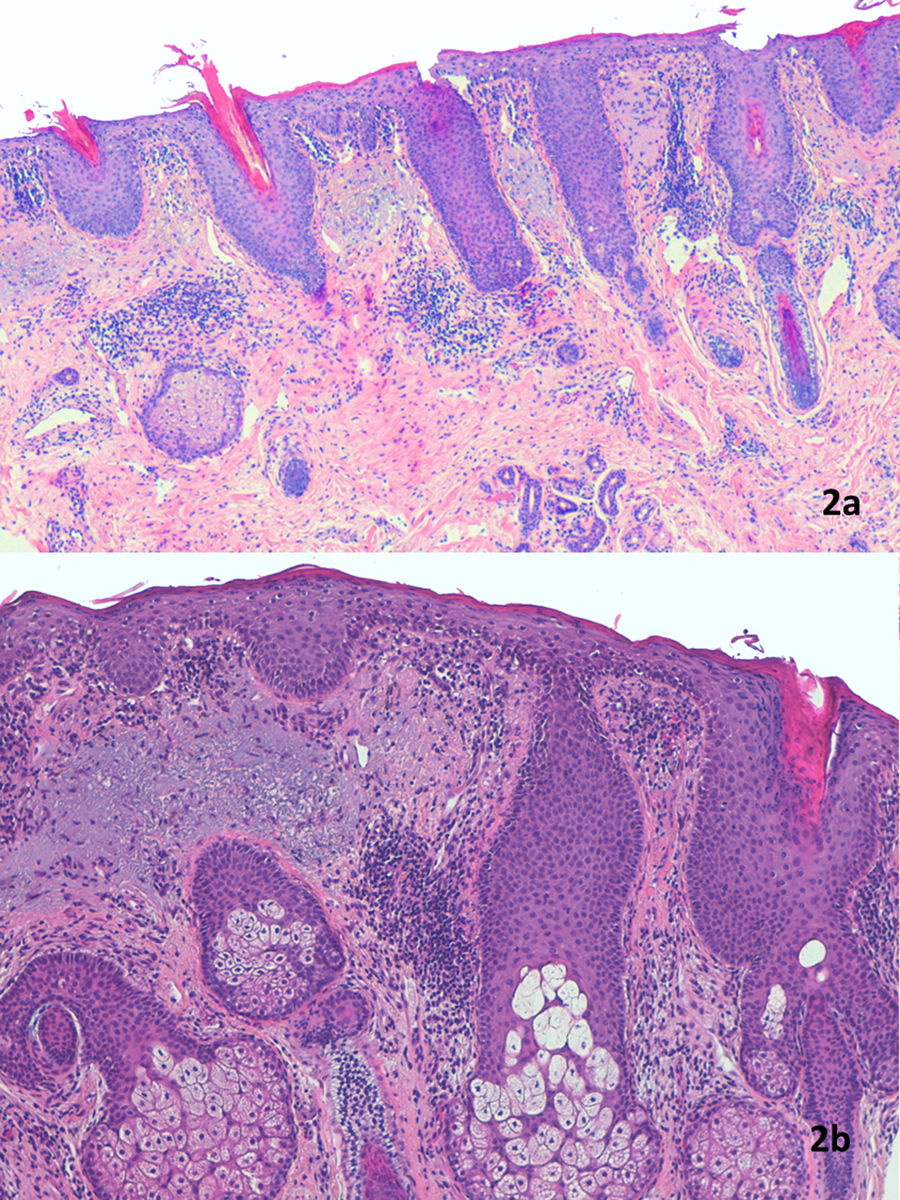
A) Epidermal hyperplasia with marked keratotic spikes in the follicular ostium and parakeratosis that is accentuated in the region surrounding the follicular infundibulum. The lymphocytic infiltrate is perivascular in the dermis. Hematoxylin–eosin (H&E) staining, magnification ×40. B) Evident epidermal acanthosis with hyperkeratosis, corneal plugs, and parakeratosis often located around follicles. Munro abscesses were not observed. Thickening of the granular layer and mild spongiosis with lymphocytic exocytosis. Perivascular lymphocytic infiltrate in the dermis. There is basophilic degeneration of collagen, without increased mucin deposition. H&E staining, magnification ×100.
FDD is characterized by macules, papules, or well-defined plaques on the face. The lesions are circular, pink-orange in color, and develop adherent scales. The clinical course is chronic and refractory to treatment. The number and size of lesions vary, and cases have been described over a wide range of ages from infancy to the sixth decade of life, usually in females.1–4
The initial description of FDD listed features that were nonspecific but suggestive of PRP.1 Histopathologic features included psoriasiform acanthosis, hyperkeratosis with parakeratosis, and follicular plugs.1 Later additions to the description included spongiosis,2,4 a perivascular lymphocytic infiltrate that spared skin adnexa,3,4 alternating orthohyperkeratosis and parakeratosis forming a checkerboard pattern with a conserved or prominent granular layer, subcorneal acantholysis, and vacuoled keratinocytes.4
One of the defining characteristics of FDD is its chronic course and resistance to numerous treatments, such as corticosteroids (topical, intralesional, and systemic), calcitriol, tacrolimus, pimecrolimus, tazarotene, imiquimod, tretinoin, topical and oral antifungal medications, hydroxychloroquine, doxycycline, methotrexate, phototherapy, and pulsed light therapy. The combination of betamethasone dipropionate with oral acitretin was reported to lead to the greatest improvement, but it still failed to achieve complete clearance.3 We report remarkably good results achieved in our patient with the combination of betamethasone and calcipotriol without acitretin.
The differential diagnosis includes other facial skin diseases such as psoriasis, lupus, seborrheic dermatitis contact dermatitis, rosacea, Demodex mite infestation, mycosis fungoides, and seborrheic pemphigus.1,4
Since the first description of this condition by Ko et al.,1 the possibility that FDD is related to PRP has been hypothesized. Clinical manifestations (salmon-colored plaques with adherent scaling), a prolonged course of disease that is resistant to treatment, and compatible histopathologic findings were the arguments put forward by these authors, who wondered whether FDD was an independent entity or a new localized form of PRP, possibly a seventh phenotype. Additional traits noted since then suggest that FDD and PRP are indeed closely related. The presence of alternating orthohyperkeratosis and parakeratosis, for example, as well as subcorneal acantholysis, and vacuoled keratinocytes are histopathologic findings that are characteristic of both processes.4,5 Gan et al.4 therefore also suggested that FDD could be an incomplete form of PRP.
PRP is one of the few skin diseases that can lead to ectropion.6,7 Classical adult onset PRP, the most severe form, might put patients at greatest risk of this complication.6 Our patient's unilateral ectropion, which was closely associated with the type and timing of the skin lesions, plus its complete resolution when the FDD was brought under control, would support a relationship between the 2 entities.
We believe that FDD could be classified as a seventh form of PRP, as originally suggested, and that this addition to the Griffiths classification system8 could well be added, just as the sixth type was added by the Spanish group of Miralles et al.9 The term FDD, therefore, would be substituted by the term “type 7 PRP” or “circumscribed facial PRP” analogous to the description of the fourth PRP type (circumscribed juvenile onset). These terms would refer to a PRP form that preferentially affects the face, is most often found in women, responds poorly to treatment in general, and tends to have a prolonged chronic course.
Conflicts of InterestThe authors declare that they have no conflicts of interest.