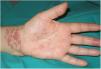
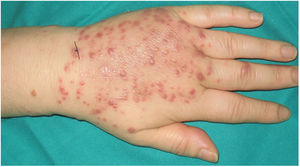
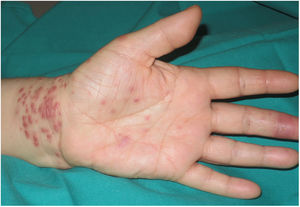
A 54-year-old woman who was being treated with subcutaneous adalimumab (40 mg/2 wk) for 1 year for rheumatoid arthritis was seen for a pruritic rash on her left hand. The rash had appeared 10 days earlier after she cut the third finger of the left hand while preparing fish in her workplace. The patient reported no previous contact with standing water (e.g. aquariums, ponds). Physical examination revealed multiple erythematous-violaceous papules of 3–4 mm on the dorsal aspect (Fig. 1) and palm of the left hand and on the wrist (Fig. 2). An erosion on the palmar aspect of the third finger of the same hand was identified by the patient as the initial injury (Fig. 2).
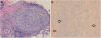
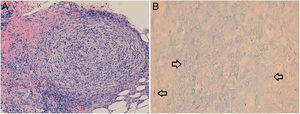
Biopsy revealed an epidermis with acanthosis and isolated apoptotic keratinocytes, band-like inflammatory infiltrate, and basal vacuolar damage. Well-defined granulomas consisting of histiocytes and lymphocytes were observed in the reticular dermis and hypodermis (Fig. 3A). Giemsa, periodic acid-Schiff, and Grocott silver staining were negative for microorganisms. Fite-Faraco staining revealed the presence of acid-alcohol resistant bacilli (Fig. 3B).
Culture of the biopsy material in Löwenstein-Jensen medium resulted in positive growth after 20 days, and Mycobacterium marinum was subsequently isolated by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS), enabling definitive diagnosis. The patient was diagnosed with a localized skin infection caused by M marinum, with a non-lymphocutaneous distribution. Antibiotic susceptibility testing showed that the microorganism was sensitive to kanamycin (high load), rifampin, ethambutol, ethionamide, cycloserine, and capreomycin, and was resistant to streptomycin, isoniazid, pyrazinamide, and paraaminosalicylic acid. The patient was initially prescribed minocycline (100 mg/12 h). After 2 months of treatment a marked improvement in the lesions was observed. However, because some active lesions persisted, antibiotic treatment was switched from minocycline to clarithromycin (500 mg/12 h). After another 2 months of treatment, the only remaining lesions were brownish macules, and antibiotic treatment was permanently discontinued. The patient experienced no subsequent recurrence after 6 months of follow-up.
Mycobacterial species other than those of the Mycobacterium tuberculosis complex and Mycobacterium leprae are known as nontuberculous mycobacteria. The incidence of infections caused by these mycobacteria is increasing dramatically, mainly due to the prevalence of AIDS and the use of immunosuppressive therapies.1 The use of tumor necrosis factor inhibitors is associated with an increased risk of tuberculosis and of infection caused by nontuberculous mycobacteria.2
The infection caused by M marinum is historically known as swimming pool or fish tank granuloma, owing to the wide distribution of this microorganism in aquatic environments, especially in stagnant water such as that found in fish ponds or swimming pools not treated with chlorine. The skin infection is acquired by contact with M marinum-contaminated water or with marine animals such as fish or crustaceans, and requires an entry site (usually minor skin trauma, which can be pre-existing or can coincide with exposure to the microorganism).
The most common clinical presentation is nodular (60% of cases), consisting of a single lesion at the inoculation site, usually affecting the upper extremities. Multiple nodular lesions occur in 35% of cases, and are generally arranged linearly following a lymphatic path from the point of inoculation, producing a classical sporotrichoid or lymphocutaneous pattern. Disseminated infections have also been described in several immunocompromised patients.3,4
Diagnosis is confirmed by culture of biopsy material. M marinum colonies are normally detected after 10 to 28 days of incubation, although cultures should be monitored for at least 6 weeks.5 In one study, only a third of acid-fast-stained samples were positive.6
A reasonable therapeutic strategy is to prescribe 2 active agents for up to 1 or 2 months after resolution of clinical signs (usually 3 or 4 months in total). For most patients, clarithromycin and ethambutol tend to provide an optimal balance of efficacy and tolerance.7
A search of the literature reveals 7 other cases of M marinum infection in patients receiving treatment with adalimumab for different diseases: rheumatoid arthritis (2), psoriasis (2), Crohn's disease (1), psoriatic arthritis (1), and ankylosing spondylitis (1).8
In conclusion, we present a case of M marinum infection with an atypical clinical presentation. This case underscores the risk of infection by nontuberculous mycobacteria in patients being treated with TNF inhibitors, and the importance of suspecting these infections, especially in individuals with a compatible exposure history.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as: Peña Merino L, Mendieta-Eckert M, Méndez Maestro I, Gardeazabal García J. Infección por Mycobacterium marinum en una paciente en tratamiento con adalimumab. Actas Dermosifiliogr. 2020;111:525–526.