Calcinosis is an infrequent complication of adult dermatomyositis (DM) and a very rare complication of paraneoplastic DM that predominantly affects areas of the limbs that are frequently exposed to pressure. Descriptions of facial calcinosis are very scarce.1 We describe the case of a patient with paraneoplastic DM and calcinosis cutis of the face.
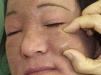
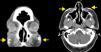
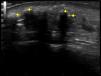
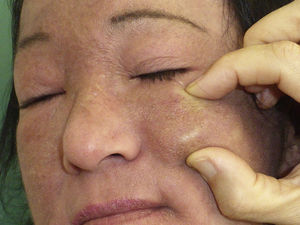
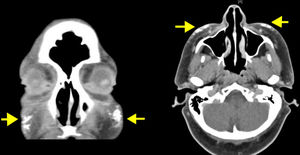
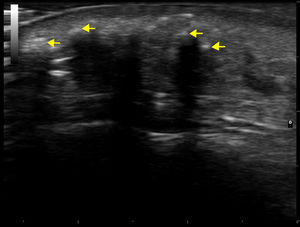
The patient was a 50-year-old woman who was being followed for DM, which had been diagnosed 8 years earlier. She had extensive DM skin lesions on the face, trunk, and limbs, and electromyography and muscle biopsy revealed involvement of the scapular and pelvic girdles. The results of the antinuclear antibody test were positive. An autoimmunity test for DM revealed positivity for anti-SAE1 (small ubiquitin-like modifier activating enzyme 1) antibody. Treatment with systemic corticosteroids, azathioprine, and methotrexate controlled the myositis but not the skin lesions, which worsened and progressed to necrotic lesions in the lumbar and gluteal regions. Mammography revealed an increase in focal density in the right retroareolar region that was difficult to evaluate; a breast biopsy was not performed until microcalcifications appeared on the breast 3 years after the onset of DM. At this point, the patient was diagnosed with grade III invasive ductal breast carcinoma. Complete remission was achieved following surgery and chemotherapy. The patient's skin condition markedly improved after breast cancer treatment. The only persisting lesions were mild poikilodermal lesions on the face and trunk that did not require immunosuppressive therapy. About 4 years after diagnosis of DM, the patient developed painful, indurated nodules, some of which were ulcerated, first on the arm and subsequently on the biopsy scar, both malar regions (Fig. 1), and the forearms, legs, and gluteal region. Radiography confirmed the presence of calcium in these locations. Craniofacial computed tomography showed thickening of the periorbital soft tissue and the presence of subcutaneous calcifications in both malar regions and the left preauricular region (Fig. 2). Skin ultrasound showed thickening of the dermis and the presence in the subcutaneous cellular tissue of hyperechoic deposits that produced posterior acoustic shadow, with no increase in vascularization evident on Doppler ultrasound (Fig. 3). Analysis of phosphocalcic metabolism revealed no findings of interest. At the time, the patient was being treated with diltiazem, colchicine, and alendronic acid (70mg/wk), which prevented progression of calcification. Several flare-ups of skin lesions, without myositis, were effectively controlled with methotrexate and cycles of intravenous immunoglobulin. The patient's breast cancer remains in complete remission and no new tumors have been detected.
Dystrophic calcinosis cutis is a relatively common complication of juvenile DM (45%–75% of cases), but is uncommon in adult DM (19%–20% of cases).1 It usually appears in patients with intense DM skin lesions, as in our patient. It is extremely rare in patients with paraneoplastic DM. No cases of calcinosis were reported in one of the largest published series of patients (N=12) with paraneoplastic DM.2 We have found reports of this association only in isolated cases in the literature.3
According to Callen and Wortmann,4 the course of paraneoplastic DM may not be dependent on the underlying tumor, but usually is. Our patient's DM did not fully resolve after treatment of the underlying tumor. However, the change in the intensity of the clinical picture and the response to treatment observed after remission of the patient's breast cancer suggest a link between DM and the tumor, which may act as a trigger of DM in predisposed individuals. Cutaneous necrosis, which was observed in our patient, is another clinical feature of DM associated with an underlying tumor.2
In DM patients, calcinosis cutis usually affects proximal parts of the limbs, muscles, and large joints.1 During the course of our patient's condition she developed calcinosis lesions on the face, in addition to the aforementioned classic locations. Isolated cases of facial calcinosis have been described in patients with systemic scleroderma,5,6 antisynthetase syndrome,7 and inflammatory acne,8 and calcinosis of the scalp has been described in 5 cases of juvenile DM.1 However, we have found no reports of facial calcinosis in cases of adult DM or paraneoplastic DM.
While anti-SAE1 antibody positivity has been associated with severe skin diseases and a higher incidence of associated cancer,9,10 no association between anti-SAE1 positivity and the development of calcinosis has been demonstrated.
In conclusion, we describe the case of a woman with paraneoplastic DM who developed dystrophic calcinosis cutis in the classic locations and on the face, a location rarely affected by this clinical form.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as: Herrero-Moyano M, Martínez-Mera C, García-García C, Dauden E. Calcinosis cutis facial en una paciente con dermatomiositis paraneoplásica. Actas Dermosifiliogr. 2018;109:760–762.