Eccrine ductal carcinoma (EDC) is a rare adnexal neoplasm that arises from the eccrine glands. It accounts for 0.01% of all skin tumors.1 The classification of the adnexal tumors is complex and the nomenclature confusing.2 Traditionally, adnexal tumors have been considered to present an aggressive behavior.
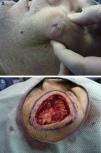
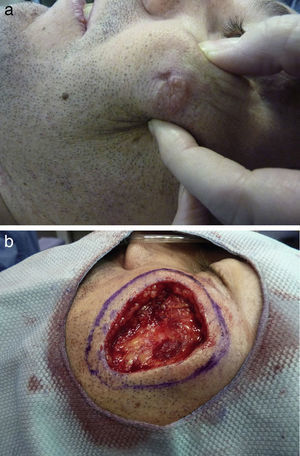
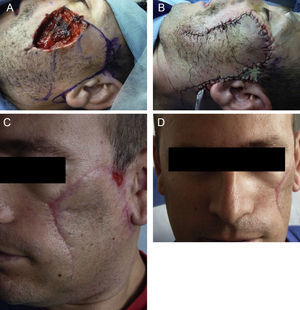
We present the case of a 37-year-old man who was referred to our department for surgical excision of a well-defined, pink-colored tumor measuring 2.8×3cm on the left cheek (Fig. 1A). The lesion, which had first appeared 2 years earlier, extended to deeper planes, and was diagnosed as an EDC. Soft-tissue ultrasound, magnetic resonance imaging, and positron emission tomography were performed and showed no deep involvement or distant spread. The excision of the lesion was extended to the muscle plane and a stage of Mohs micrographic surgery was performed for routine processing (Fig. 1B). Histology of the surgical specimen showed an intensely sclerotic epithelial tumor that infiltrated to the muscle (Fig. 2A). Structures with a ductal appearance and areas of squamous differentiation were observed within the tumor (Fig. 2B). Immunohistochemistry was positive for low molecular weight (Fig. 2C) and high molecular weight (Fig. 2D) cytokeratins and CD10 (Fig. 2E), and the cell proliferation index was low (Fig. 2F). No tumor nests were detected in the specimen sent for routine study. Surgical reconstruction was performed in a second operation using a rotation flap from the cheek and a Burow graft (Fig. 3, A and B), with good cosmetic and functional results (Fig. 3, C and D). A year after surgery the patient remains disease free.
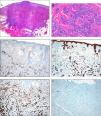
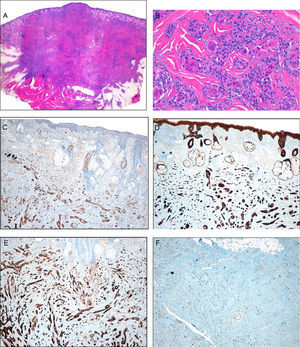
Histology. A, Low magnification showing a dense, nonencapsulated infiltrate that reaches the muscle layer. Hematoxylin-eosin (H&E), original magnification ×4. B, Detail of the fibrous stroma, with the presence of glandular lumens and cells grouped in cords. Note the squamous differentiation. H&E, original magnification ×40. C, Immunohistochemistry. Positivity for low molecular weight cytokeratins. Original magnification ×10. D, Immunohistochemistry. Positivity for high molecular weight cytokeratins. Original magnification ×10. E, Immunohistochemistry. Positivity for CD10. Original magnification, ×10. F, Immunohistochemistry. Stain for Ki67. Note the low cell proliferation index. Original magnification, ×10.
EDC is the most common type of adnexal tumor arising from the eccrine glands1 and is included in the group of tumors of uncertain behavior.3 Because of the small number of cases published, the prognosis remains unknown; the tumor appears to be locally aggressive but with a low probability of metastatic spread. Avraham et al.4 analyzed more than 200 adnexal tumors with eccrine differentiation, and the majority were found to be in stage i. Clinically, these are slow-growing, erythematous-violaceous tumors that may ulcerate. The clinical and histological differential diagnosis is broad. EDCs are more common in middle-aged and older men and arise preferentially on the scalp, trunk, and limbs.1
The clinical differential diagnosis includes basal cell carcinoma, squamous cell carcinoma, skin metastases, cutaneous lymphomas, and amelanotic melanoma. The diagnosis is histological. The architecture of these tumors resembles that of ductal carcinoma of the breast, with nests and cords of cells, a variable degree of glandular differentiation, and infiltration of the adjacent tissues.5 EDCs typically stain positive for carcinoembryonic antigen, epithelial membrane antigen, and cytokeratins 5/6.2 Isolated cases with squamous differentiation have been reported1,5,6; the origin of these tumors is controversial and it has been suggested that they may be squamous cell carcinomas arising in the tubules.7 This confusion is probably due to the complexity of their diagnosis, which will have led to underdiagnosis. The histological differential diagnosis includes other adnexal tumors, squamous cell carcinoma in those cases with extensive squamous differentiation, and a possible collision between squamous cell carcinoma and EDC.6
Imaging studies for staging have not been standardized, though screening for local invasion and distant spread is recommended. Because EDC is such a rare tumor, it has been included in the TNM staging for squamous cell carcinoma.
Recommended treatment is by surgical excision of the tumor with negative margins, either by radical surgical excision or using Mohs micrographic surgery.5,6 Although margin width has not been defined,7 margins should be broad because of the tendency to underestimate the size of the tumor.8 Margins between 1 and 2cm have been sought in the majority of cases. Mohs micrographic surgery has been used with good results in other adnexal tumors,9 although reports have not specified whether fresh-tissue or fixed-tissue techniques were used. Some authors use 2mm layers of resection,7 others use 5mm,9 and some even support excising an additional layer after obtaining a tumor-free result.10 Comparing the 2 techniques, no local recurrences have been detected in the few cases treated by Mohs micrographic surgery, whereas recurrence rates of up to 70% have been reported after wide surgical excision,9 although the follow-up periods have been short. With respect to adjuvant treatment, these tumors have been shown to respond poorly to radiation therapy4 and chemotherapy. Adjuvant radiation therapy has not been shown to increase survival.
In conclusion, EDC is probably a less aggressive tumor than has been thought, but local recurrence is common. The squamous variant appears to be more common than has been reported in the literature and is probably underdiagnosed. In our opinion, Mohs micrographic surgery is a good therapeutic option for EDC, but diagnostic-therapeutic algorithms still need to be developed.
Please cite this article as: Lorente Luna M, Jiménez Blázquez E, Sánchez Herreros C, Cuevas Santos J. Carcinoma ductal ecrino: papel de la cirugía de Mohs y revisión de la literatura. Actas Dermosifiliogr. 2016;107:73–76.