Exposure to ultraviolet radiation (UVR) is the leading risk factor for developing cutaneous melanoma and the only modifiable factor known to date. Multiple mutations linked to melanoma have also been reported, the most common somatic mutation being a V600E substitution in BRAF. The main sources of UVR exposure are sunlight and tanning booths. As early as the 1980s, numerous epidemiological studies had found a strong association between a history of sunburn and the risk of developing skin cancer. Surprisingly however, and contrary to what one might expect given the results of laboratory experiments, several population studies have linked the use of sunscreens with an increased risk of developing melanoma1 and with a higher number of melanocytic nevi in children and adolescents.2 One explanation for these seemingly contradictory findings could be that, in people who intentionally expose themselves to sunlight, sunscreen use delays the onset of burns and, therefore, increases the duration of exposure to UVR.3 However, both the mechanism by which UVR induces melanoma and the role of sunscreen use in preventing the disease has, for decades, remained unclear.
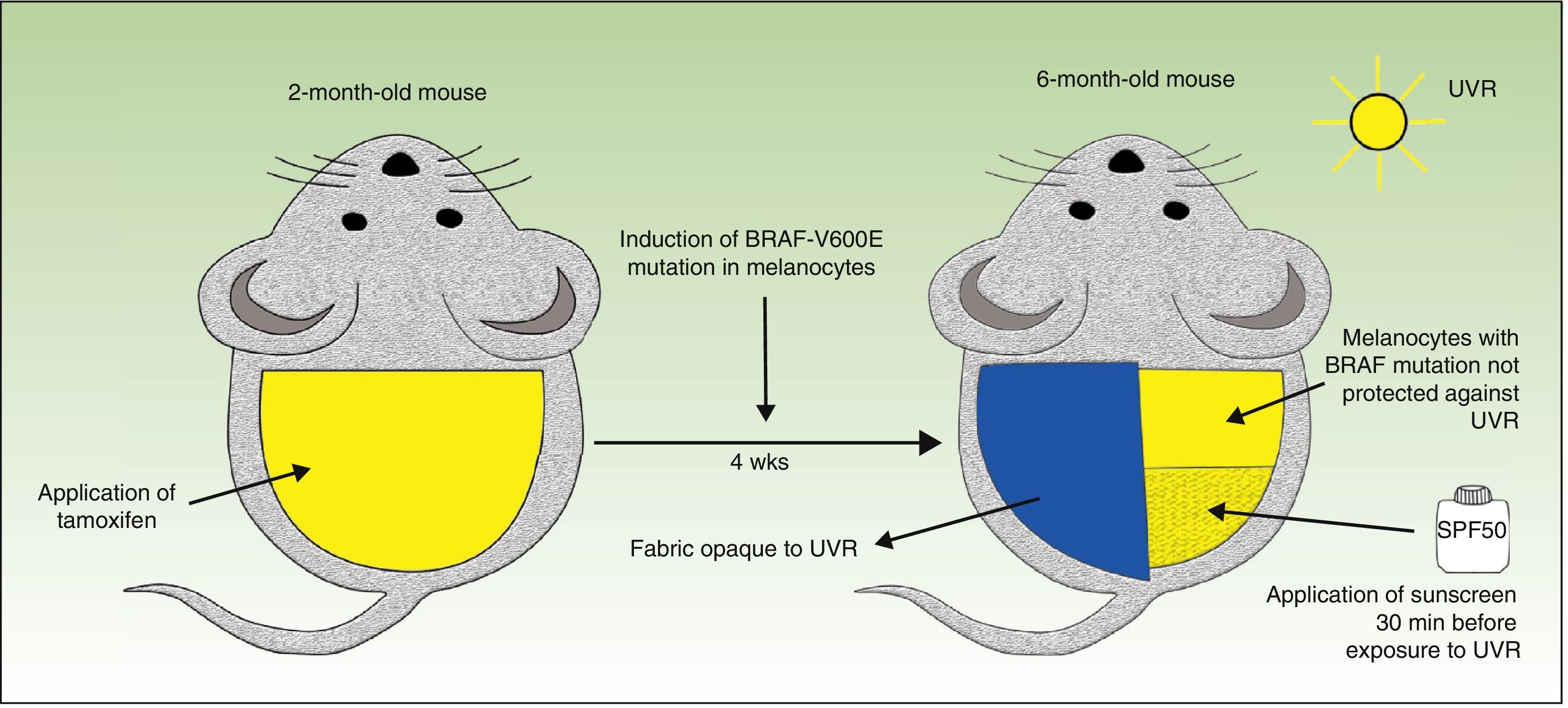
The journal Nature recently published a fascinating study carried out by a group of British researchers who investigated the effect of UVR on melanomagenesis in a mouse model expressing BRAF (V600E) in melanocytes.4 They used conditional knockout mice (genetically engineered using the Cre-loxP system); the mice carried the mutated BRAF V600E gene but its expression was suppressed until activated. It is possible, using Cre-LoxP technology, to repress the normal gene and activate the expression of the mutated gene at a specific point in the study. The Cre protein is an enzyme, and its expression can be induced by several different substances, including tetracycline and tamoxifen. Topical application of tamoxifen to the shaved back of the animal induced the expression of the BRAF V600E mutation only in the area where the activating substance was applied. One month later, half of the area in which the mutated gene had been activated was covered with a fabric opaque to UVR and the unprotected half of the back was exposed weekly for 6 months to UV-A or UV-B radiation (160mJ/cm−2) from a broad spectrum lamp. To investigate the effect of sunscreen in preventing melanoma the investigators applied an FDA-recommended, broad-spectrum (UV-B or UV-A) sunscreen product with an SPF of 50 (2.2 mg/cm−2), 30min before exposure to UVR (Fig. 1).
The authors of the study made several observations of great interest and clinical significance. First, these low doses of UVR (which induced mild erythema) induced an increase in the size and development of new nevi only in the areas of skin with the BRAF mutation; this phenomenon was not observed in the areas without mutation. Second, all the mice with the BRAF mutation exposed to UVR developed melanomas, while the same exposure of skin without the mutation produced no increase in melanoma incidence, indicating that UVR accelerates the BRAF-driven melanomagenesis process. Another finding of great significance was that the application of sunscreen blocks the short-term effects of UVR (induction of Trp53, apoptosis of epidermal keratinocytes, epidermal hypertrophy, and thickening of the dermis); however, in mice carrying the BRAF mutation, it delayed rather than completely prevented the development of melanoma. In addition, the authors demonstrated that topical application of the sunscreen reduces, but does not completely eliminate, the DNA damage induced by UVR.
In conclusion, the study cited has demonstrated that the TP53/Trp53 mutations induced by UVR accelerate the process of BRAF-driven melanomagenesis. Furthermore, the results support the use of other photoprotection strategies in addition to sunscreen in at risk individuals who carry the BRAF mutation.
The authors wish to acknowledge the invaluable help of K. Kokoriev in developing the figure.
Please cite this article as: Pastushenko I, Gilaberte-Calzada Y. Do Sunscreens Eliminate the Risk of Melanoma?. Actas Dermosifiliogr. 2015;106:837–838.