We report the case of a 60-year-old man who was receiving immunosuppressive therapy for a bilateral lung transplant and presented with a crusted, violaceous plaque on the left hand. Based on histopathology and microbiological culture the patient was diagnosed with infection by Alternaria species. Treatment with itraconazole led to complete resolution of the skin lesion. Forty months later he developed four reddish, nodular, skin lesions on the left leg. Analysis of a biopsy from one of these lesions using histopathologic and molecular techniques identified a mold that shared 98% homology with a strain of Alternaria triticina. Alternaria species belong to a group of dematiaceous fungi that cause opportunistic infections in humans. The incidence of these infections is increasing, mainly in transplant centers. To the best of our knowledge, this is the first reported case of a human infection caused by A. triticina.
Presentamos el caso de un hombre de 60 años en tratamiento inmunosupresor por un trasplante pulmonar bilateral que presentó una placa violácea costrosa en la mano. Se diagnosticó de infección por Alternaria spp mediante estudio histopatológico y cultivo microbiológico. El tratamiento con itraconazol condujo a la resolución completa de la lesión cutánea. Cuarenta meses más tarde el paciente desarrolló 4 lesiones nodulares rojizas en la pierna izquierda. Las muestras obtenidas de una de las lesiones fueron estudiadas mediante histopatología y técnicas moleculares. La secuencia de ADN que se obtuvo del germen causal mostraba una homología del 98% con una cepa de Alternaria triticina. Las especies de Alternaria forman parte del grupo de hongos que componen las feohifomicosis susceptibles de causar infecciones oportunísticas humanas. La incidencia de estas infecciones está aumentando, sobre todo en centros de trasplante. De acuerdo con nuestra información el presente caso es el primero en el que A. triticina es causante de una infección humana.
The incidence of phaeohyphomycoses, opportunistic infections caused by dematiaceous molds,1 has increased in recent years. Fungi belonging to the genus Alternaria are increasingly associated with opportunistic mycosis in immunocompromised hosts.2 The most common clinical manifestations are cutaneous and subcutaneous infections, as well as oculomycosis, rhinosinusitis, and onychomycosis.3 The most important risk factors for cutaneous and subcutaneous infections are Cushing syndrome and solid organ transplant.3 We report a case of cutaneous infection by Alternaria triticina in a bilateral lung transplant patient. To the best of our knowledge, this is the first case of human infection with this kind of organism reported to date.
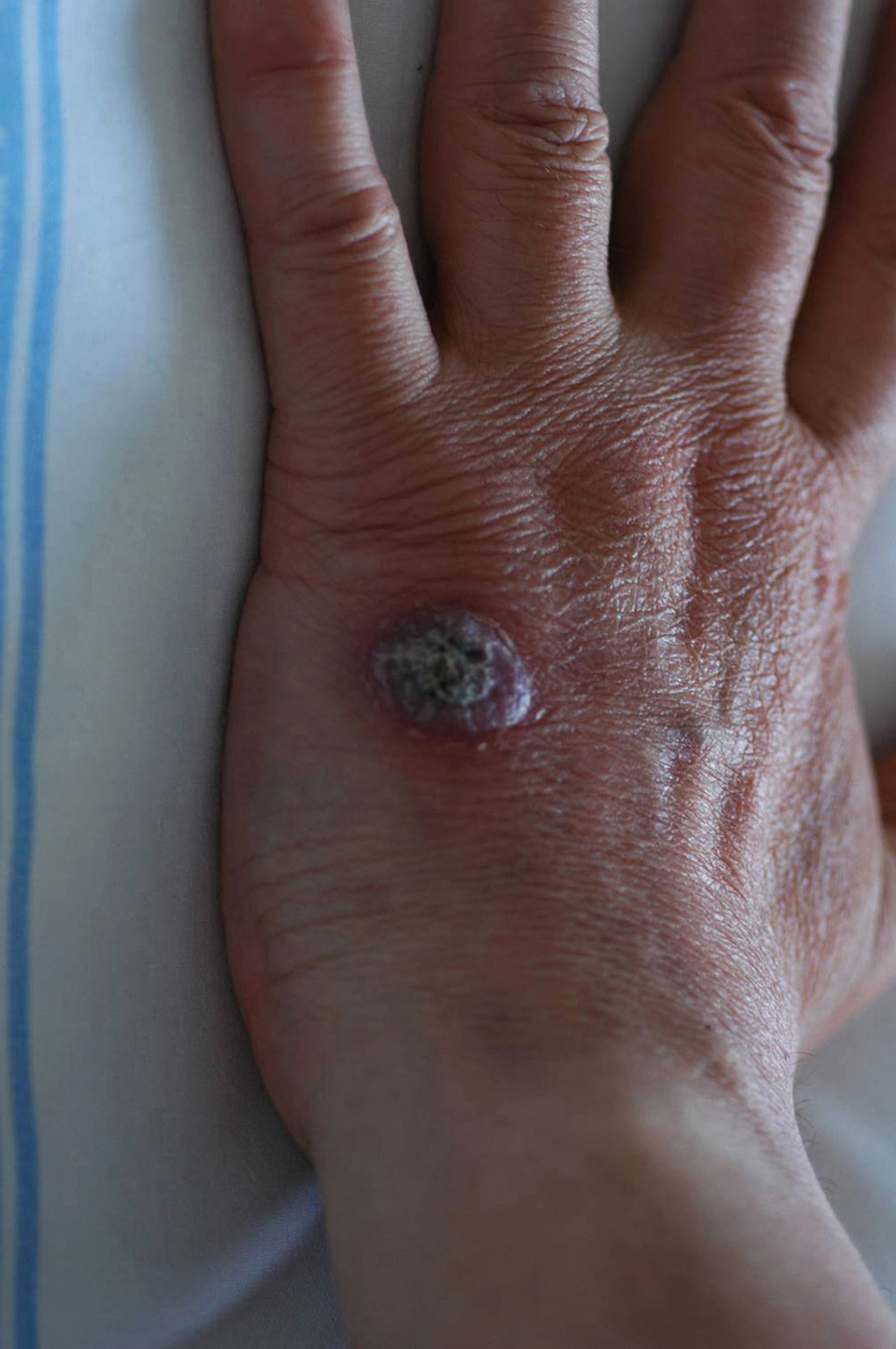
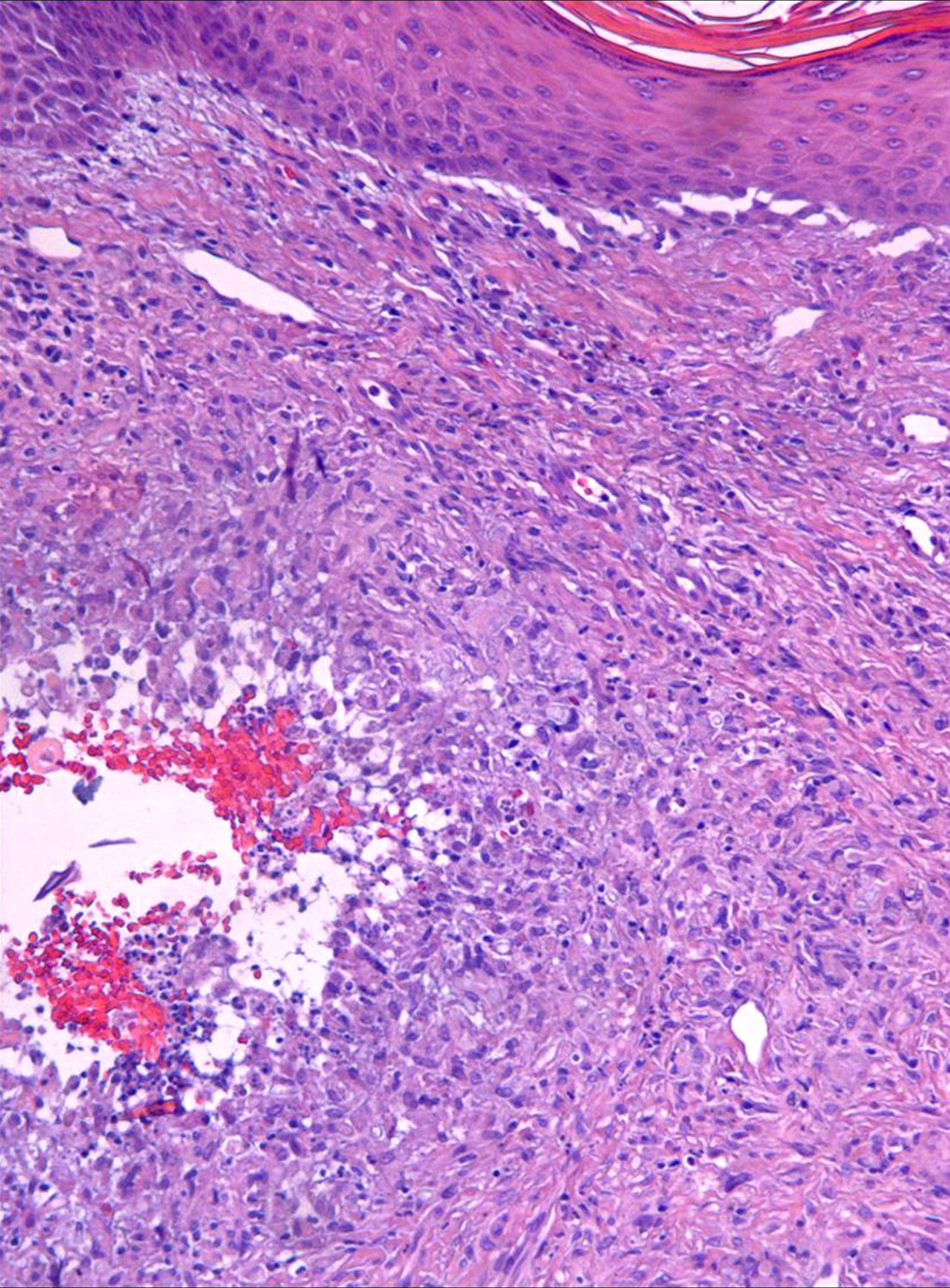
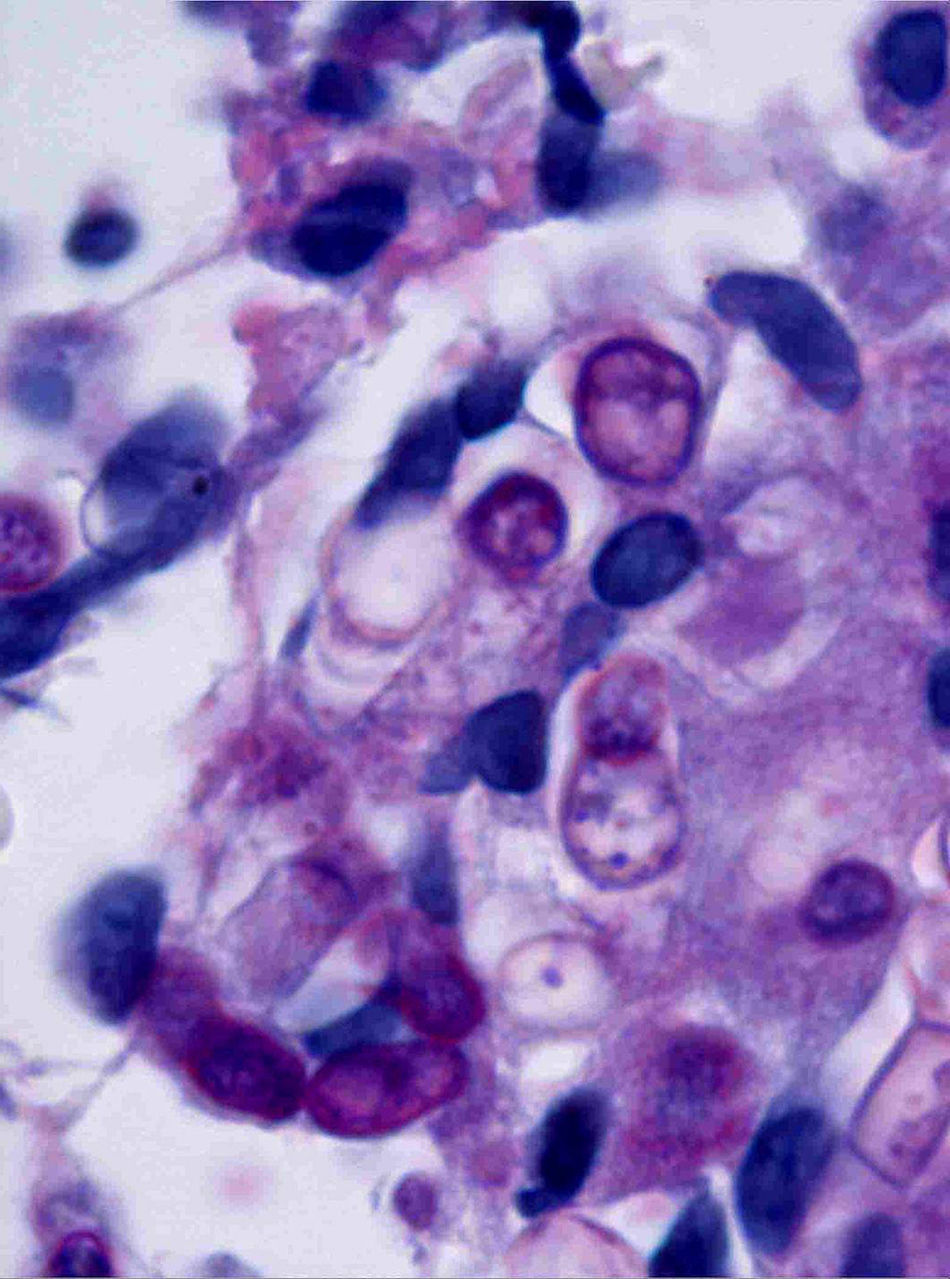
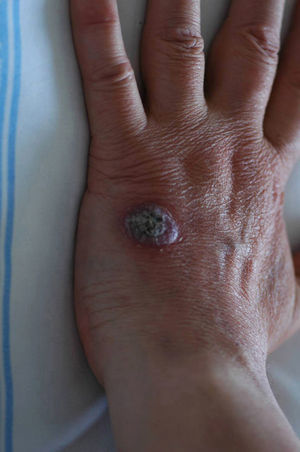
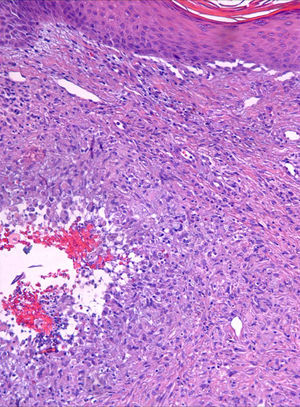
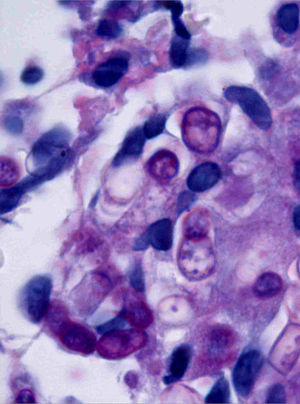
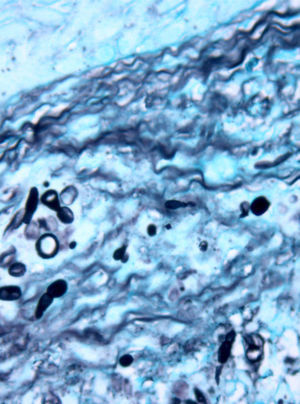
Case descriptionA 60-year-old man presented with a crusted, purple plaque on the left hand (Fig. 1). The lesion was painless and the patient could recall no history of injury at that site. He had undergone bilateral lung transplantation 4 months previously and was receiving immunosuppressive therapy (500mg mycophenolate mofetil, 1mg tacrolimus, and 15mg prednisone daily). A skin biopsy was taken for histopathological examination and microbiological culture. Histopathology revealed suppurative, granulomatous inflammation in the dermis and hypodermis. The lesion was composed of lymphocytes, epithelioid macrophages, multinucleated giant cells, and aggregates of neutrophils in the center of the granulomas (Fig. 2). Periodic acid-Schiff (PAS) stain revealed round-shaped, occasionally septate, fungal structures within the cytoplasm of some giant cells that were associated with extracellular fungal hyphae (Fig. 3).
Fungal cultures were performed on Sabouraud dextrose agar. Velvety, yellowish-white colonies with an olive-green reverse, suggesting the presence of dematiaceous fungi, developed after 12 days of incubation. Microscopic examination of these colonies revealed brown, septate hyphae without conidia. The diagnosis was infection with Alternaria species. The patient was treated with 100mg itraconazole twice a day for 6 months, leading to complete resolution of the lesion.
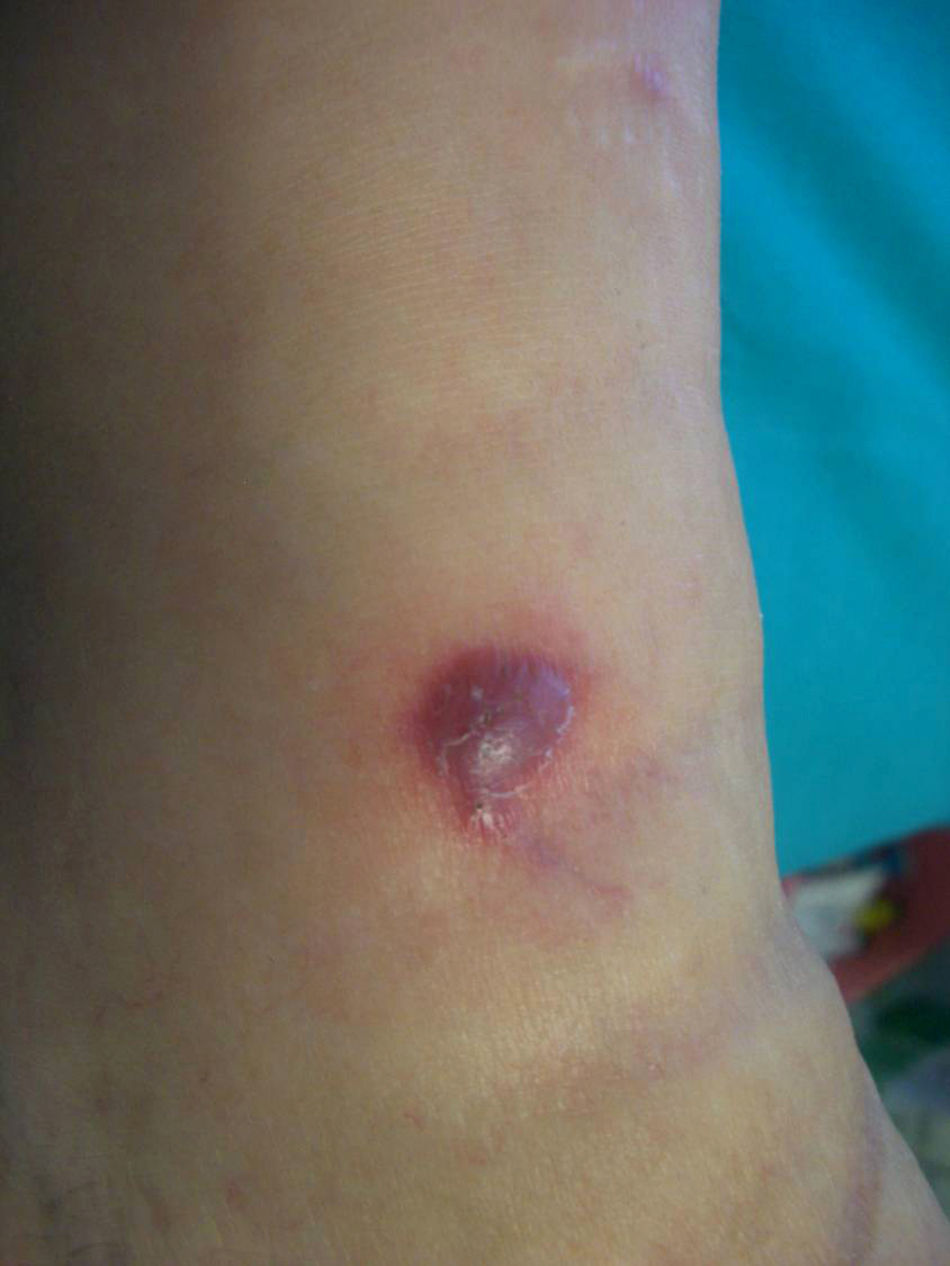
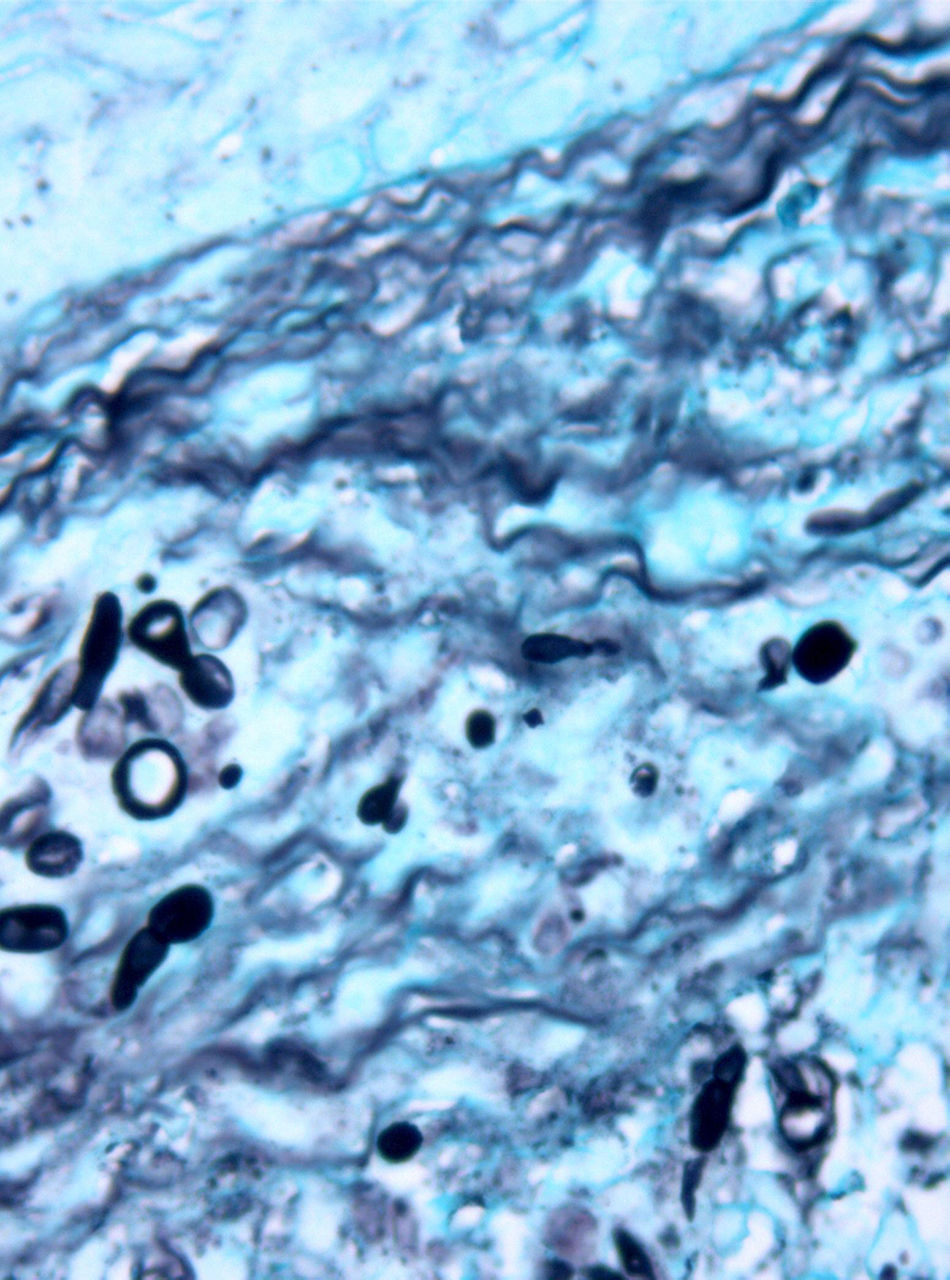
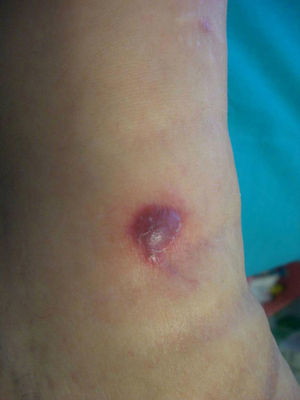
Forty months later the patient developed four reddish, nodular skin lesions on the lower left leg (Fig. 4). A biopsy of one of these lesions was performed, and histopathology revealed suppurative, granulomatous inflammation in the dermis. Grocott’ methenamine silver nitrate staining showed diffusely distributed fungal elements (Fig. 5). Samples were subcultured on Sabouraud dextrose agar and produced fast-growing, fungal colonies with an olive-green undersurface. Direct species differentiation was hampered by the absence of conidia, and so molecular techniques were used. DNA was extracted from the biopsy and the Internal Transcribed Spacer (ITS) 1–2 region, which includes the 5.8S ribosomal DNA gene, was amplified using universal primers ITS1-4.4 The sequence obtained (552 base pairs) was compared with those in the GenBank DNA database (www.ncbi.nlm.nih.gov). This sequence showed 98% homology with a strain of A. triticina. The colonies were then transferred to Czapek agar (Difco) and analyzed by microscope, which revealed oval, brown conidia with horizontal and longitudinal septa.
The strain was also sent for analysis to the Instituto Nacional de Salud Carlos III, Madrid, a center with an extensive microorganism database. Analyses performed there using molecular techniques confirmed our own findings. The ribosomal RNA gene sequences of ITS1-5.8S and ITS-2 regions of the clinical isolate described here (isolate HUMV 59512) have been deposited in GenBank (accession number GU318225).
The patient was treated twice per day for 10 months with 200mg oral voriconazole, which resulted in complete resolution of all cutaneous lesions. No recurrences have been reported after 25 months of follow-up.
DiscussionAlternaria is a very large and complex genus that encompasses hundreds of species, only a few of which have been involved in human or animal infections. The most common species implicated in human disease are Alternaria alternata and Alternaria infectoria, although in many reported cases of alternariosis the species has not been determined.3
A. triticina, the species isolated from the cultures in the present case, has not been previously described as a human pathogen. To the best of our knowledge, this is the first such case reported in the literature.
A. triticina is a plant pathogen that causes leaf blight on wheat. It was first identified in India in 1962 and continues to cause significant wheat crop yield losses on the Indian subcontinent.5 The disease causes necrotic leaf lesions and in severe cases seeds become shriveled. Alternaria triticina can be distinguished from other Alternaria species by agarose gel electrophoresis.6
Most cases of human Alternaria infection involve immunocompromised individuals. Cutaneous alternariosis has been described in organ transplant recipients and in patients with Cushing syndrome, lymphoproliferative disorders, and AIDS.3,7,8
Two routes of cutaneous infection have been proposed: exogenous, following traumatic inoculation with fungal elements (e.g., through injury with a plant spine) or after colonization of pathologically altered skin; and endogenous, caused by inhalation of fungal conidia and subsequent systemic spread, which eventually results in secondary cutaneous involvement.7
Cutaneous alternariosis can range from local skin lesions to invasive and disseminated infection. Clinical features include verruciform, eczematous, or ulcerating plaques, as well as vegetating or nodular lesions.7 Skin lesions can be single or multiple and usually develop on exposed sites or bone prominences such as the feet, knees, legs, backs of the hands, forearms, and less frequently, the face. Histopathologically, cutaneous Alternaria infections usually manifest with noncaseating granulomas and chronic inflammation in the dermis, often with microabscesses. Ulceration or pseudoepitheliomatous hyperplasia of the epidermis may be observed. Melanized hyphae and round-to-oval refractile structures within the histiocytes are common features of fungal infections, including phaeohyphomycoses. Samaniego González et al.8 proposed that these refractile structures, which may appear septate depending on type of section analyzed, correspond to muriform conidia in culture, and serve as a diagnostic clue in cases of cutaneous alternariosis. However, culture is essential for the correct identification of Alternaria species.
Treatment of cutaneous alternariosis is controversial. Attenuation of immunosuppression or surgical excision of localized lesions can be sufficient to resolve the infection.3 Many antifungal drugs have been used with varying degrees of success and itraconazole is considered the treatment of choice.9,10 However, the dose and treatment duration are not well standardized. In some cases, surgery is necessary as an adjuvant to oral therapy.11 Relapses can occur even after long-term treatment. In these types of cases, voriconazole and liposomal amphotericin B may be an alternative treatment option.
The genus Alternaria belongs to a group of dematiaceous fungi that cause phaeohyphomycoses and are increasingly associated with opportunistic mycoses in immunocompromised hosts. A high index of suspicion is warranted when examining unusual skin lesions on exposed parts of the limbs of transplant recipients. The diagnosis of cutaneous alternariosis requires a positive tissue culture and histological identification. Early detection and treatment is important to avoid disseminated cutaneous lesions.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this investigation.
Confidentiality of dataThe authors declare that they have followed the protocols of their hospitals concerning the publication of patient data, and that all the patients included in this study were appropriately informed and gave their written informed consent.
Right to privacy and informed consentThe authors declare that no private patient data appears in this article.
Conflict of interestThe authors declare no conflict of interest.
We thank Manuel Cuenca Estrella and Emilia Mellado of the Instituto Nacional de Salud Carlos III in Madrid for confirmation of the identity of the fungus.