Platelet-rich plasma (PRP) is used as an adjuvant in the treatment of chronic ulcers of the lower extremity and has shown particularly promising results in the case of neuropathic ulcers. There has been less research, however, into its use in venous and hypertensive ulcers. Our aim was to assess the safety and feasibility of using PRP in the treatment of chronic ulcers of the lower extremity and to evaluate its potential benefits in directed healing.
Material and methodsWe prospectively selected 11 patients with nonischemic ulcers of the lower extremity that had been present for at least 6 weeks. PRP was injected subcutaneously into the perilesional tissue and applied topically in 4 sessions held at 1-week intervals. We assessed quality of life (SF-12 questionnaire), pain (visual analog scale), and the circumference of the ulcer before and after treatment.
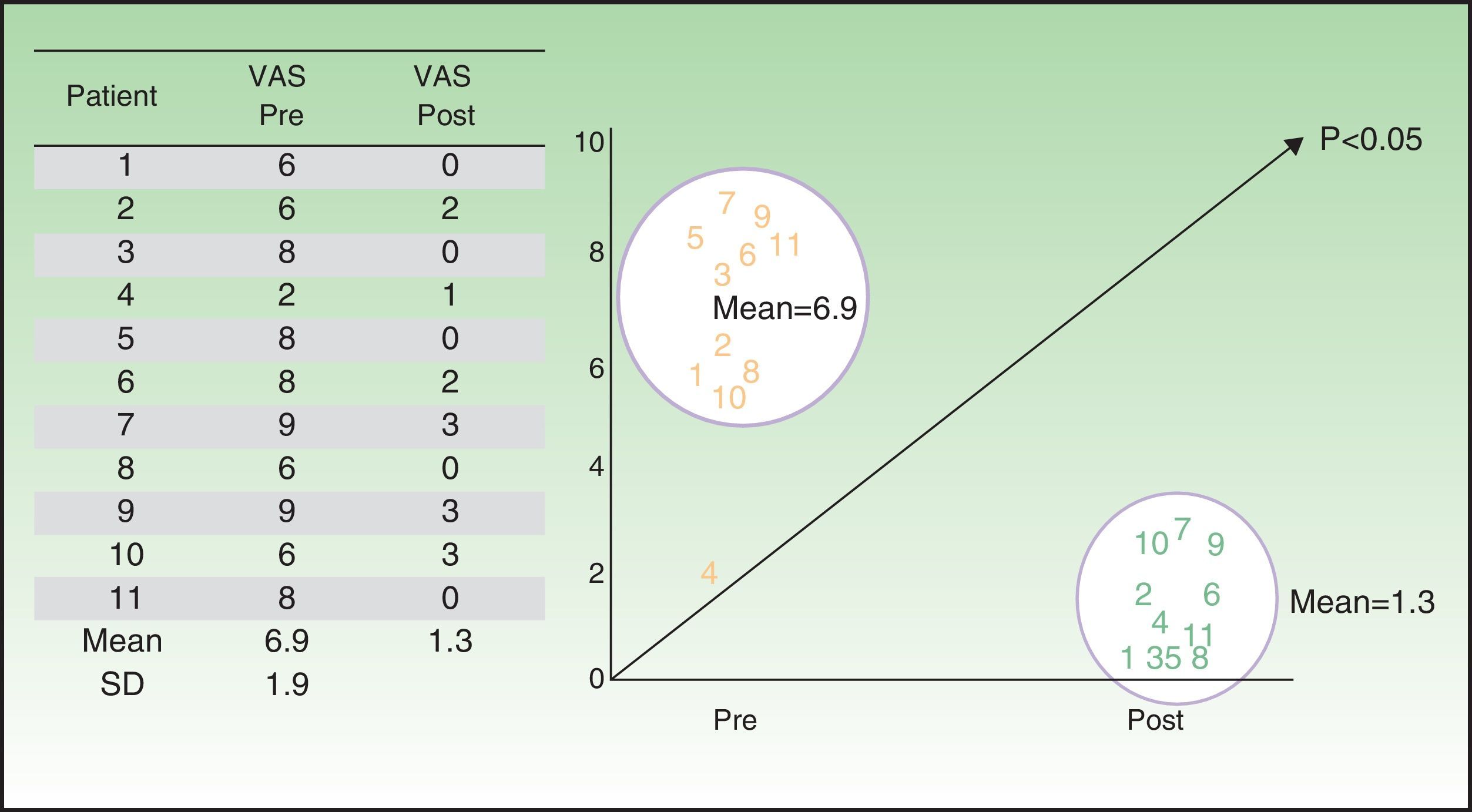
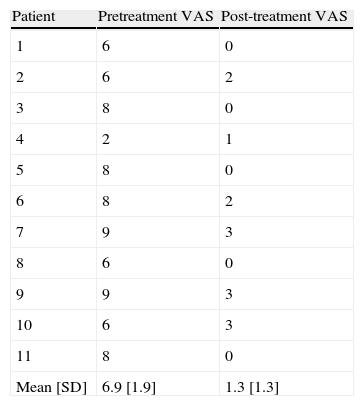
ResultsThere was a predominance of women (8/11, 73%), and venous ulcers (7/11, 64%) were more common than hypertensive ulcers (4/11, 36%). The median age of the patients was 79 years and the median time since onset of the ulcer was 17 months (range, 6-108 months). We observed a significant reduction in pain (P<.05) and a significant improvement in the physical and mental components of the SF-12 (P<.05). The mean reduction in ulcer size was 60%, and complete healing was achieved in 5 cases. No adverse effects were observed.
ConclusionThe local application of PRP is a valuable and practical procedure that promotes the healing of chronic ulcers of the lower extremity; it can improve patient quality of life and is particularly effective in local pain relief.
El plasma rico en factores plaquetarios (PRP) ha sido utilizado como terapia adyuvante en el tratamiento de úlceras crónicas de miembros inferiores (UCMI), con resultados particularmente esperanzadores en las úlceras neuropáticas. Sin embargo, su uso en úlceras venosas e hipertensivas ha sido menos estudiado. Nuestro objetivo es valorar la seguridad y factibilidad del uso del PRP en UCMI y estimar sus beneficios en la cicatrización dirigida.
Material y métodosSe trata de un estudio prospectivo. Fueron seleccionados 11 pacientes con úlceras en miembros inferiores de más de 6 semanas de evolución, de etiología no isquémica. Se administró PRP en inyección subcutánea perilesional y aplicación tópica, en 4 sesiones separadas por una semana. Se suministraron cuestionarios de calidad de vida (SF-12), se registró el dolor (escala visual analógica) y el perímetro de la úlcera antes y después de su aplicación.
ResultadosHubo predominancia de mujeres (8/11=73%) y de úlceras venosas (7/11=64%) sobre las hipertensivas (4/11=36%). La mediana de edad fue de 79 años y el tiempo medio de evolución de la úlcera de 17 meses (6-108 meses). Se evidenció una disminución estadísticamente significativa del dolor (p<0,05) y mejoría mental y física de la calidad de vida (p<0,05). Se evidenció una reducción media en el área de las úlceras del 60%, con 5 cicatrizaciones completas. No se registraron eventos adversos.
ConclusiónLa aplicación local de PRP en úlceras crónicas de miembros inferiores representa una herramienta válida, de uso práctico en la cicatrización dirigida, que puede mejorar la calidad de vida de estos pacientes, siendo particularmente útil en el control del dolor local.
Chronic ulceration of the lower extremities refers to a group of diseases that, through different pathophysiological mechanisms that alter the normal physiological healing process, have the common outcome of the appearance and tendency to progression of a solution of continuity of the skin below the level of the knees for a period of 6 weeks or longer.1
The treatment of chronic leg ulcers, whatever their etiology, consists fundamentally of the treatment of the primary cause (arterial or venous disease, neuropathy, etc.) and simultaneous local treatment of the ulcer, with the application of dressings, drugs, or factors that together promote restoration of the normal physiological healing process.
Platelet-rich plasma (PRP) is the plasma fraction derived from autologous blood that, after processing, has a higher platelet concentration than at baseline.2 PRP has been used as adjuvant therapy for the treatment of chronic leg ulcers, and the results in neuropathic ulcers in diabetic patients have been very promising.3–5
It serves both as a sealant and as a medium for drug delivery.6 In addition to its notable anti-inflammatory effect,2,3the contents of the α granules are released to act locally to stimulate the initiation of healing (platelet-derived growth factors, transforming growth factorβ, platelet factor4, interleukin-1, platelet-derived angiogenic factor, vascular endothelial growth factor, epidermal growth factor, platelet-derived endothelial growth factor, epithelial cell growth factor, insulin-like growth factor, osteocalcin, osteonectin, fibrinogen, vitronectin, thrombospondin-1).7 Furthermore, some studies have shown that PRP has antimicrobial activity against Escherichia coli, Staphylococcus aureus, Candida albicans and Cryptococcus neoformans.8,9
The objective of the this prospective study was to describe an outpatient protocol for the use of plasma rich in platelet-derived growth factors for the topical treatment of chronic leg ulcers of nonischemic etiology in order to evaluate the feasibility and viability of its routine use and to estimate its potential benefit in targeted healing.
As we describe a protocol for outpatient use, ulcers of ischemic etiology were excluded as those patients are admitted for semi-urgent revascularization procedures.
Materials and MethodsPatients were selected from the targeted-healing outpatient clinic between December 2011 and January 2013.
Patients included in the study were those who, after at least 6 weeks of supervised treatment at our center, presented no clinical improvement or reduction in the surface area of the ulcer (determined photographically). Prestudy outpatient treatment consisted of the application of occlusive moist dressings, as long as the attending physician considered the risk of infection to be sufficiently low.
After checking that patients satisfied the inclusion criteria and presented none of the exclusion criteria (Table 1), a maximum of 4 local applications of PRP were performed at 1-week intervals (Table 2). Preparation of the autologous plasma to obtain the fraction rich in platelet-derived factors to be employed in the treatment was the accepted method standardized by the scientific research committee at our hospital. In the case of venous ulcers, single-layer compression bandages were also used.
Selection Criteria.
| Inclusion |
| Age>18 y |
| Walks without assistance from other persons and does not require orthoses to perform daily activities |
| Below-knee ulcer present for more than 6 weeks, of venous, neuropathic, or hypertensive etiology |
| Written authorization by the patient |
| Pedal/posterior tibial pulses present or ankle-brachial index>0.8 |
| Exclusion |
| Ulcers of ischemic etiology |
| Ulcers present for more than 6 weeks but showing signs of healing (on photographic comparison with the previous week) |
Technique.
| Extraction of 4 tubes of blood from the patient in the clinic (each tube contained approximately 8mL of blood) |
| Processing in installations specialized in the management of cell cultures and other advanced therapies |
| Centrifugation of the blood at 5000rpm for 8min |
| Extraction of the platelet-rich fraction of the plasma, situated immediately above the red cell layer (approximately 1mL of plasma) |
| Lysis of the platelet membrane by the addition of 10% calcium carbonate: 0.05mL permL of plasma |
| Warming of the plasma obtained to 37°C for 5min |
| Conventional wound care with topical anesthesia and debridement as appropriate (cleaning the base of the ulcer with saline and chlorhexidine soap and removal of slough) |
| Photograph of the ulcer with reference ruler |
| Injection of two thirds of the volume of PRP into the base of the ulcer and topical application of the remaining third to the surface of the ulcer |
| Dressing with petrolatum gauze |
| Bandage and heel protection as necessary |
After completing the study treatment, individualized conventional wound care was performed in each case according to standard clinical practice (as explained above).
The following information was gathered: personal and demographic details, clinical data, etiology of the ulcer and time since onset, cardiovascular risk factors, and past medical and surgical history. Data were stored in an Excel spreadsheet and were analyzed by parameter inference using the Epidat 3.1 software package.
Clinical improvement was defined as follows:
- 1
Improvement in the SF-12 quality of life survey (Spanish version).
- 2
Reduction of the area of the treated ulcer, measured on digital photographs taken throughout follow-up and evaluated using a specific computer program (Adobe Acrobat).
- 3
A statistically significant reduction in the score on the visual analog scale (VAS) for pain before and after treatment.
The SF-12 quality of life survey (in Spanish, see additional material) was administered and the pain level, measured on the VAS for pain, was recorded before the first session and immediately after the final session.
Clinical and photographic follow-up of the ulcers was performed for 8 weeks after the final application of PRP.
ResultsEleven patients satisfied the inclusion criteria. The large majority of patients were women and were hypertensive (Table 3). The median duration of active ulceration was 17 months (range, 7-108 months).
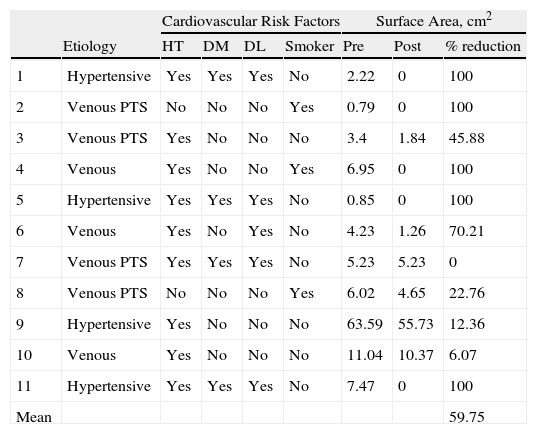
The most common etiology was venous insufficiency, followed by hypertension (Table 4). The ulcers were most frequently situated in the area of the medial malleolus (54%), followed by the lateral malleolus (27%); bilateral (medial and lateral) malleolar ulcers were present in 18% of cases. None of the patients with ulcers of venous etiology underwent surgery prior to, during, or after of the application of the PRP.
No adverse events of any type were detected related to the application of PRP.
In the 8 weeks after the final application of PRP, complete healing was observed in 5 ulcers and the mean ulcer surface area decreased by 59.8% (Table 5).
Reduction in Ulcer Surface Area After the Application of PRP.
| Cardiovascular Risk Factors | Surface Area, cm2 | |||||||
| Etiology | HT | DM | DL | Smoker | Pre | Post | % reduction | |
| 1 | Hypertensive | Yes | Yes | Yes | No | 2.22 | 0 | 100 |
| 2 | Venous PTS | No | No | No | Yes | 0.79 | 0 | 100 |
| 3 | Venous PTS | Yes | No | No | No | 3.4 | 1.84 | 45.88 |
| 4 | Venous | Yes | No | No | Yes | 6.95 | 0 | 100 |
| 5 | Hypertensive | Yes | Yes | Yes | No | 0.85 | 0 | 100 |
| 6 | Venous | Yes | No | Yes | No | 4.23 | 1.26 | 70.21 |
| 7 | Venous PTS | Yes | Yes | Yes | No | 5.23 | 5.23 | 0 |
| 8 | Venous PTS | No | No | No | Yes | 6.02 | 4.65 | 22.76 |
| 9 | Hypertensive | Yes | No | No | No | 63.59 | 55.73 | 12.36 |
| 10 | Venous | Yes | No | No | No | 11.04 | 10.37 | 6.07 |
| 11 | Hypertensive | Yes | Yes | Yes | No | 7.47 | 0 | 100 |
| Mean | 59.75 | |||||||
Abbreviations: DL, dyslipidemia; DM, diabetes mellitus; HT, hypertension; PRP, platelet-rich plasma; Venous PTS, venous insufficiency secondary to posthrombotic syndrome.
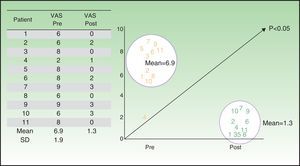
The patient-perceived ulcer-related pain showed a statistically significant reduction (P<0.05) between baseline (mean VAS score, 6.7) and end of treatment (mean VAS score, 1.3) (Table 6 and Fig. 1).
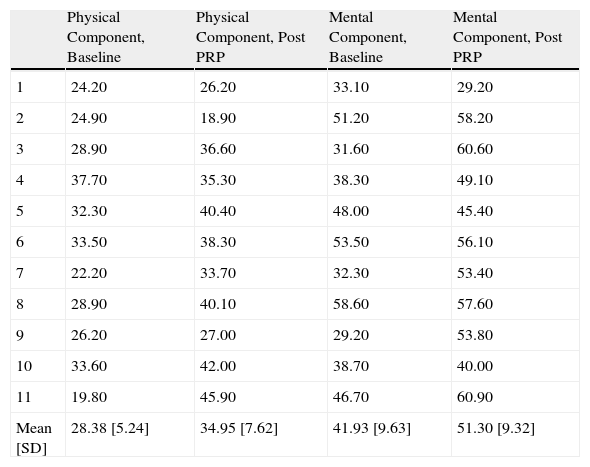
Likewise, we observed a statistically significant improvement (P<0.05) in the mental and physical quality of life components reported by the patients after the application of PRP (Table 7).
Quality of Life Score at Baseline and After PRP Therapy. gr1.
| Physical Component, Baseline | Physical Component, Post PRP | Mental Component, Baseline | Mental Component, Post PRP | |
| 1 | 24.20 | 26.20 | 33.10 | 29.20 |
| 2 | 24.90 | 18.90 | 51.20 | 58.20 |
| 3 | 28.90 | 36.60 | 31.60 | 60.60 |
| 4 | 37.70 | 35.30 | 38.30 | 49.10 |
| 5 | 32.30 | 40.40 | 48.00 | 45.40 |
| 6 | 33.50 | 38.30 | 53.50 | 56.10 |
| 7 | 22.20 | 33.70 | 32.30 | 53.40 |
| 8 | 28.90 | 40.10 | 58.60 | 57.60 |
| 9 | 26.20 | 27.00 | 29.20 | 53.80 |
| 10 | 33.60 | 42.00 | 38.70 | 40.00 |
| 11 | 19.80 | 45.90 | 46.70 | 60.90 |
| Mean [SD] | 28.38 [5.24] | 34.95 [7.62] | 41.93 [9.63] | 51.30 [9.32] |
Abbreviation: PRP, platelet-rich plasma.
Chronic lower-leg ulceration is a major public health problem. Its high prevalence and chronic nature lead to an elevated cost to the health system and a marked deterioration in the quality of life of affected individuals. Its treatment is complex; scientific evidence is conflicting and endorses various alternatives, most of which are valid and have acceptable results.
Numerous studies have demonstrated the safety of the application of PRP and support and promote its use in a wide variety of clinical applications, including chronic ulceration of the lower extremities of various etiologies.3–5,10,11
Our series of patients presented a significant reduction in ulcer surface area (59.8%), and we achieved a considerable percentage of complete closure of the ulcer. Other studies have reported the same effect.12–14 One randomized clinical trial reported a 72.9% reduction in ulcer surface area in patients treated with PRP, compared with a 21.5% reduction in the control group.12
No adverse events were recorded in our study. A review of the literature revealed that, in general, the application of PRP would appear to be relatively safe. All the clinical trials we found that recorded these events reported no statistically significant differences between the study group and control group. In fact, some of those studies reported a lower rate of events in patients treated with PRP.12–14
The use of PRP had a beneficial effect on local pain control in the majority of our patients. We observed a significant reduction in the scores on the World Health Organization VAS for pain between baseline and after application of the treatment. We have found no other studies that have specifically evaluated this variable during the treatment of chronic ulcers with PRP. In similar studies in which PRP has been used to treat acute ulcers and surgical wounds, a statistically significant reduction was observed in the intravenous analgesic requirements of the patients undergoing this therapy.15
We also detected a statistically significant improvement in the mental and physical quality of life components between baseline and after the application of PRP. In the literature we reviewed, we found no studies that have evaluated quality of life related to the application of PRP. Although our data are insufficient to make a definitive statement, it would appear logical to infer that this improvement may be related to the reduction in pain felt by the patients, as this pain (particularly in a chronic setting) leads to negative physical and psychological phenomena.
ConclusionsIt is necessary to study strategies that combine different therapeutic elements in order to accelerate ulcer healing and improve the patients’ quality of life. One of the tools available to achieve this objective, PRP, is a useful and practical treatment for targeted healing of chronic ulceration of the lower extremities.
There is a need for further well-designed studies with a larger number of patients that include PRP as part of a strategy for targeted healing and that compare this with traditional strategies in order to determine the usefulness and benefit of the application of PRP in daily clinical practice.
Ethical DisclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this investigation.
Confidentiality of dataThe authors declare that they followed their hospital's regulations regarding the publication of patient information and that written informed consent for voluntary participation was obtained for all patients.
Right to privacy and informed consentThe authors declare that no private patient data are disclosed in this article.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
We would like to thank Casto Barbero and Marco Fernández-Beltrán, nurses of the Wound Care Clinic in Vascular Surgery Outpatients in our hospital, who were responsible for performing the targeted healing method before, during, and after the application of PRP.
Please cite this article as: Salazar-Álvarez AE, Riera-del-Moral LF, García-Arranz M, Álvarez-García J, Concepción-Rodriguez NA, Riera-de-Cubas L. Uso de plasma rico en plaquetas para cicatrización de úlceras crónicas de miembrs inferiores. Actas Dermosifiliogr. 2014;105:597–604.