New biological drugs are highly valuable tools for the treatment of patients with moderate-to-severe psoriasis.1 However, due to decreases in their long-term efficacy, these treatments can pose compliance problems, which can affect survival and treatment persistence.2 Given that the objective of psoriasis treatment is effective long-term control of skin manifestations,3,4 it is very important to standardize the criteria to evaluate drug persistence, which is mainly determined by efficacy, safety, side effects, ease of administration, and patient satisfaction.5 The main problem with clinical trials that have evaluated these therapies is that they apply very strict inclusion criteria and have short follow-up periods, making their results difficult to extrapolate to clinical practice.6–10
To evaluate patient persistence with biologic therapies for moderate-to-severe psoriasis in clinical practice, we conducted an opinion survey of patients with this disease, placing special emphasis on treatment with biological drugs. The objective was to improve knowledge of the disease from the patient’s point of view and to optimize treatment.
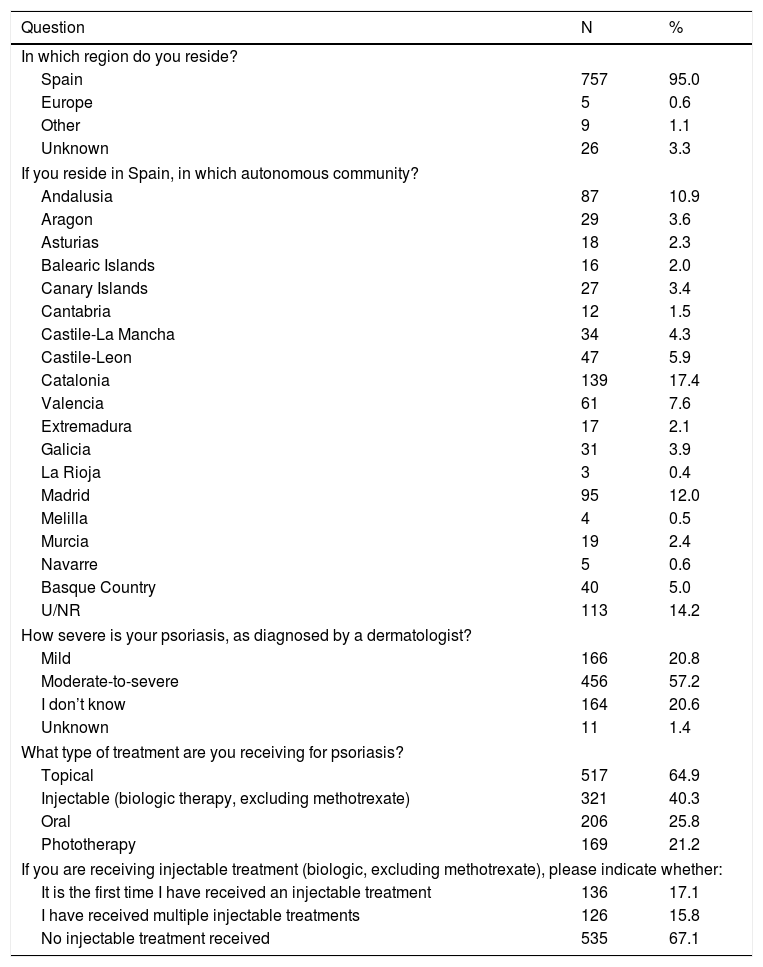
The questionnaire was created and distributed by Acción Psoriasis after validation by a group of psoriasis experts, and was carried out in 2 phases. The first phase consisted of an anonymous 5-question questionnaire about the characteristics of the patients, the type of psoriasis they had, and any treatments they were receiving. The questionnaire was sent electronically to 797 patients in the Acción Psoriasis database. Table 1 shows the characteristics of the patients who responded to this first questionnaire. Most of the patients were Spanish nationals (95%) and were diagnosed with moderate-to-severe psoriasis (57.2%). The most commonly prescribed treatment was topical (64.9%), followed by injectable (biologics) (40.3%), oral (25.8%), and phototherapy (21.2%). Among patients who received multiple concomitant treatments, the most frequent combination was topical treatment together with phototherapy (15.3%), followed by topical and oral treatment (4.8%), and the combination of topical treatment, injectable (biologic) treatment, and phototherapy (1.6%).
Results of the First Questionnaire (N=797).
| Question | N | % |
|---|---|---|
| In which region do you reside? | ||
| Spain | 757 | 95.0 |
| Europe | 5 | 0.6 |
| Other | 9 | 1.1 |
| Unknown | 26 | 3.3 |
| If you reside in Spain, in which autonomous community? | ||
| Andalusia | 87 | 10.9 |
| Aragon | 29 | 3.6 |
| Asturias | 18 | 2.3 |
| Balearic Islands | 16 | 2.0 |
| Canary Islands | 27 | 3.4 |
| Cantabria | 12 | 1.5 |
| Castile-La Mancha | 34 | 4.3 |
| Castile-Leon | 47 | 5.9 |
| Catalonia | 139 | 17.4 |
| Valencia | 61 | 7.6 |
| Extremadura | 17 | 2.1 |
| Galicia | 31 | 3.9 |
| La Rioja | 3 | 0.4 |
| Madrid | 95 | 12.0 |
| Melilla | 4 | 0.5 |
| Murcia | 19 | 2.4 |
| Navarre | 5 | 0.6 |
| Basque Country | 40 | 5.0 |
| U/NR | 113 | 14.2 |
| How severe is your psoriasis, as diagnosed by a dermatologist? | ||
| Mild | 166 | 20.8 |
| Moderate-to-severe | 456 | 57.2 |
| I don’t know | 164 | 20.6 |
| Unknown | 11 | 1.4 |
| What type of treatment are you receiving for psoriasis? | ||
| Topical | 517 | 64.9 |
| Injectable (biologic therapy, excluding methotrexate) | 321 | 40.3 |
| Oral | 206 | 25.8 |
| Phototherapy | 169 | 21.2 |
| If you are receiving injectable treatment (biologic, excluding methotrexate), please indicate whether: | ||
| It is the first time I have received an injectable treatment | 136 | 17.1 |
| I have received multiple injectable treatments | 126 | 15.8 |
| No injectable treatment received | 535 | 67.1 |
aA given patient could receive several types of treatment.
Abbreviation: U/NR, unknown/no response.
From the patients who responded to the first questionnaire, we selected Spanish nationals (to ensure that they were beneficiaries of the national health system) from all autonomous communities who had moderate-to-severe psoriasis and had received biologic therapy. Of the 797 patients surveyed, 231 (29%) fulfilled these criteria.
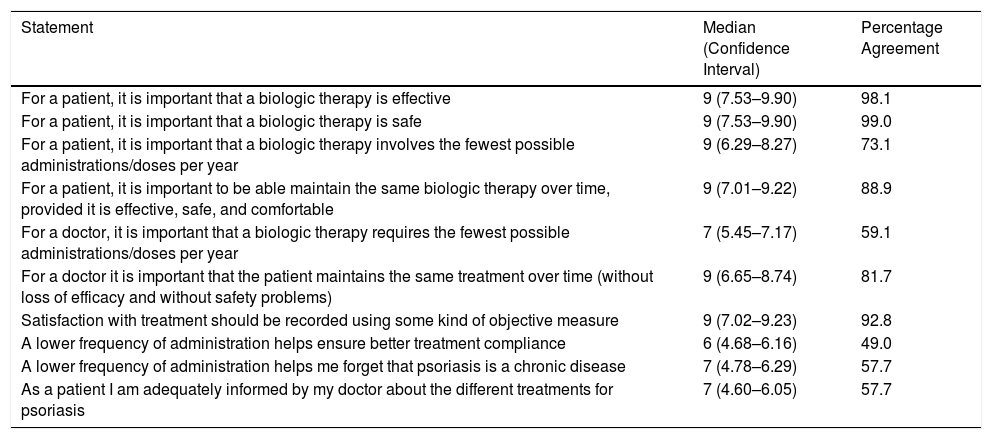
In the second phase, a second questionnaire was sent to the patients who met the aforementioned selection criteria. The questionnaire consisted of 10 specific statements about biologic therapy. Participants were required to indicate their degree of agreement with each statement using a 9-point Likert scale: disagree, 1–3; neither agree nor disagree, 6–8; agree, 7–9. The questionnaire was completed correctly by 208 of the 231 selected patients (90.0%). The results of this questionnaire are shown in Table 2.
Results of the Second Questionnaire (N=208).
| Statement | Median (Confidence Interval) | Percentage Agreement |
|---|---|---|
| For a patient, it is important that a biologic therapy is effective | 9 (7.53–9.90) | 98.1 |
| For a patient, it is important that a biologic therapy is safe | 9 (7.53–9.90) | 99.0 |
| For a patient, it is important that a biologic therapy involves the fewest possible administrations/doses per year | 9 (6.29–8.27) | 73.1 |
| For a patient, it is important to be able maintain the same biologic therapy over time, provided it is effective, safe, and comfortable | 9 (7.01–9.22) | 88.9 |
| For a doctor, it is important that a biologic therapy requires the fewest possible administrations/doses per year | 7 (5.45–7.17) | 59.1 |
| For a doctor it is important that the patient maintains the same treatment over time (without loss of efficacy and without safety problems) | 9 (6.65–8.74) | 81.7 |
| Satisfaction with treatment should be recorded using some kind of objective measure | 9 (7.02–9.23) | 92.8 |
| A lower frequency of administration helps ensure better treatment compliance | 6 (4.68–6.16) | 49.0 |
| A lower frequency of administration helps me forget that psoriasis is a chronic disease | 7 (4.78–6.29) | 57.7 |
| As a patient I am adequately informed by my doctor about the different treatments for psoriasis | 7 (4.60–6.05) | 57.7 |
aPercentage of patients who responded with a score of 7–9 on the Likert scale.
Most of those surveyed attributed great importance to safety (99.0%), efficacy (98.1%), being able to maintain the same treatment over time (88.9%), and administration in the fewest possible doses (73.1%). A high percentage of patients felt that their satisfaction with treatment should be recorded in their clinical history using some objective measure (92.8%). The frequency of treatment administration was considered a determinant of treatment adherence by 49.0% of participants, and 57.7% felt that a lower frequency of administration helped them forget that psoriasis is a chronic process. Just over half of those surveyed (57.7%) felt that they had received sufficient information from their doctor about the different treatment options for psoriasis.
The results of this questionnaire shed light on the opinions of patients with moderate-to-severe psoriasis on persistence with biologic therapy in clinical practice, complementing data previously obtained in randomized clinical trials. Practically all patients surveyed felt that to ensure persistence with biologic therapies it is essential to use the lowest frequency of administration possible and to take into account treatment efficacy, safety, and satisfaction. There was less consensus about reducing the frequency of administration to improve treatment adherence or perception of the disease, possibly because this is more relevant to the physician than the patient. Not all respondents agreed that they received sufficient information from their doctor. It is thus extremely important to improve doctor-patient communication regarding available treatments and to facilitate shared decision-making.
FundingThis work was funded by Janssen-Cilag (Spain).
Conflicts of InterestLP has received fees for consulting and/or speaking, and for participation in clinical trials, from AbbVie, Almirall, Amgen, Baxalta, Biogen, Boehringer Ingelheim, Celgene, Gebro, Janssen-Cilag, Leo Pharma, Lilly, Merck-Serono, MSD, Mylan, Novartis, Pfizer, Regeneron, Roche, Sandoz, Samsung Bioepis, Sanofi, and UCB. IA and SS are employees of Janssen-Cilag, Spain. SA has no conflicts of interest to report.
The authors would like to thank Acción Psoriasis for their participation in the preparation and distribution of the questionnaires among the patients in their database, as well as Dr. Fernando Sánchez Barbero and Luzan 5 Health Consulting for their help in preparing the manuscript.
Please cite this article as: Puig L, Alarcón I, Sulleiro S, Alfonso S. Encuesta de opinión a pacientes con psoriasis moderada-grave sobre la persistencia de los tratamientos biológicos. Actas Dermosifiliogr. 2020. https://doi.org/10.1016/j.ad.2020.01.001