Approximately 1% of the general population have venous or arterial lower limb ulcers. These lesions can be treated with biological skin substitutes such as cadaver skin or tissue-engineered skin equivalents, but treatment fails in 25% of cases, resulting in pain and loss of patient autonomy, as well as increased morbidity and health care costs. In the treatment of corneal ulcers, amniotic membrane has been shown to have antimicrobial and bacteriostatic properties, and to protect the wound without eliciting an immune response. The same properties have been reported in the treatment of burns and postthrombotic ulcers.
ObjectivesTo assess the effectiveness of amniotic membrane transplantation in the treatment of refractory chronic leg ulcers.
Patients and methodsAmniotic membrane was grafted onto 4 refractory ulcers in 3 patients. The mean time required for partial and complete reepithelialization was calculated by measuring the wound area at weeks 0, 4, 8, 12, and 16. Pain intensity was assessed at the same intervals using a visual analog scale.
ResultsComplete wound reepithelialization was achieved for 1 ulcer by week 8; in the other 3 cases, there was a 50% reduction in size compared to baseline. At week 16, the mean reduction in wound size for the 4 ulcers was 81.93%. The corresponding reduction in pain intensity was 86.6%. No adverse effects were observed.
ConclusionsAmniotic membrane transplantation might be an effective alternative for the treatment of refractory chronic vascular ulcers on the lower limbs.
Las úlceras vasculares (venosas y arteriales) de las extremidades inferiores afectan aproximadamente al 1% de la población. Para su tratamiento pueden utilizarse equivalentes cutáneos vivos procedentes de donantes cadavéricos o desarrollados mediante bioingeniería tisular. Un 25% de las úlceras no se resuelven, lo que conlleva dolor y pérdida de autonomía para los pacientes, además de un aumento de la morbilidad y de los costes sanitarios. La membrana amniótica aplicada en úlceras corneales ha demostrado poseer propiedades antimicrobianas y bacteriostáticas, y ser capaz de proteger la herida sin inducir respuesta inmunológica. En úlceras postrombóticas y quemaduras también se han descrito estas mismas cualidades.
ObjetivosValorar la eficacia de la aplicación del injerto de membrana amniótica en las úlceras vasculares crónicas refractarias de extremidades inferiores.
Pacientes y métodosSe aplicó el injerto de membrana amniótica en cuatro úlceras refractarias de tres pacientes. Se calculó el tiempo medio de epitelización total o parcial de la lesión mediante el registro del área y se valoró el dolor mediante escala visual analógica, en las semanas 0, 4, 8, 12 y 16.
ResultadosEn la semana 8 se observó la epitelización completa de una úlcera y la reducción del 50% de las otras tres. En la semana 16 la reducción del área fue de media un 81,93% en los cuatro casos. La intensidad del dolor percibido se redujo un 86,6%. No se detectaron efectos adversos.
ConclusionesLa membrana amniótica puede ser una alternativa eficaz en el tratamiento de las úlceras vasculares crónicas refractarias de extremidades inferiores.
Venous and arterial lower limb ulcers are very common and affect approximately 1% of the general population.1 The standard treatment, which consists of cleaning, debridement, and application of dressings, achieves cure rates of 65% to 85%. About 25% of these ulcers become chronic, resulting in pain, loss of patient autonomy, increased morbidity, and increased health care costs.1,2 The biological, synthetic, and biosynthetic dressings developed to promote healing include human cadaver skin and tissue-engineered skin equivalents,1–3 but these are not routinely used because of their high cost.3,4
Amniotic membrane is currently used in ophthalmology to reconstruct the corneal surface in cases of corneal ulcer. In this application amniotic membrane has shown protective, antimicrobial, and bacteriostatic properties without inducing immunological reactions because of its low expression of the HLA-A, B, C and DR antigens, its capacity to inhibit proteases and to stimulate inflammatory cells into rapid apoptosis, and the (as yet not fully demonstrated) presence of antibacterial factors in amniotic fluid.5 The use of human amniotic membrane in dermatology as a dressing for wounds and burns dates back to the early 20th century. With the improvement in sterilization techniques in the late 20th century, the possibility of using amniotic membrane to treat vascular ulcers was considered.6–11 In 2007 Mermet et al.12 performed the first prospective study of long-standing vascular ulcers refractory to standard treatment, demonstrating that amniotic membrane significantly reduced ulcer size, pain intensity, and reepithelialization time through its capacity to promote growth, adhesion, and differentiation of epithelial cells and to prevent their apoptosis.
Subsequently, Pesteil et al.8,13 confirmed these results. The benefits obtained by these authors and those reported in the literature for other dermatological conditions8–18 raised the possibility of using amniotic membrane to treat vascular ulcers refractory to standard treatment. In conjunction with the tissue bank at our hospital, which provided us with the amniotic membrane, we set up a prospective, uncontrolled, observational pilot study based on a series of clinical cases in order to investigate the effectiveness of amniotic membrane transplantation in the treatment of refractory chronic leg ulcers.
Patients and MethodsThe study was approved by the clinical research ethics committee at Hospital Clínic de Barcelona. Following the signing of informed consent, amniotic membrane was grafted onto 4 refractory chronic vascular ulcers on the lower limbs of 3 patients. The patients had venous and arterial lower limb ulcers that had appeared over 6 months earlier. After treatment for 1 year with hydrocolloid and polyurethane foam dressings and even, in 1 case, with the failed application of an autograft, the patients had acheived reepithelialization of less than 0.4cm2 per month and had negative serology for hepatitis B and C viruses and human immunodeficiency virus.
Ulcers with bacterial infection or presence of fibrotic or necrotic tissue were excluded.
The amniotic membrane was obtained and processed at the tissue bank of the Transplant Services Foundation of Hospital Clínic de Barcelona. The criteria for selecting amniotic membrane donors were based on the standards of the Spanish Association of Tissue Banks. Specific exclusion criteria were uncontrolled pregnancy, an abnormal obstetric history, symptoms of infection in the newborn, delivery before 34 weeks’ gestation, and membrane rupture more than 12hours before delivery.14,15 The donors gave written informed consent for the donation and use of the amniotic membrane and were screened antepartum for hepatitis B and C viruses and human immunodeficiency virus. The placentas were obtained from elective cesarean deliveries to avoid structural defects and contamination of the placenta by the vaginal flora, chlamydia, herpes, or other infectious agents present in the birth canal.15
After donation the placenta was washed with physiological saline to remove traces of blood and kept at 4°C. A smear was taken immediately after collection of the placenta and placed for no longer than 24hours in RPMI medium containing 50mg/mL amphotericin, 50mg/mL penicillin, and 50μg/mL streptomycin. In the laboratory, the amniotic membrane was identified and separated from the placenta by dissection in a laminar flow chamber; fragments with no infiltrated blood, tearing, or other abnormalities were considered suitable. The stromal side of the membrane was located, placed on the filter paper used as a support, and trimmed. The fragments were placed in labeled containers and a sample of each was taken for microbiological tests.
Amniotic membrane transplantation was performed on an outpatient basis in the day hospital of the dermatology department of Hospital Clínic de Barcelona.
The causes of the ulcers were treated where possible and ulcer size was measured using the Visitrak digital system. Bacterial infection was ruled out according to the clinical appearance of the ulcer, the surrounding skin, and the exudate. Pain was assessed using a visual analog scale, where 0 represented no pain and 10 represented the worst pain imaginable. Data were collected at weeks 0, 4, 8, 12, and 16.
The preparation of the ulcers included sponging with soap, mechanical debridement of fibrinous debris or slough with a scalpel or curette when necessary, and a compression bandage in cases without contraindications. The membrane preservation solution was removed by washing with physiological saline and the membrane was applied directly onto the ulcer bed. The graft was immediately covered with petrolatum-impregnated gauze and a secondary polyurethane foam dressing. Patients were confined to bed for 2hours and then allowed to do moderate activity for the next 5 days.
In all cases a single amniotic membrane transplantation was performed and 2 follow-up visits were held per week to assess progress and to perform standard wound care.
The Stat Crunch computer program was used to record ulcer size and pain intensity prior to amniotic membrane transplantation and 4, 8, 12, and 16 weeks after treatment. The data were analyzed descriptively using absolute and relative frequencies, means (SD), and medians (range).
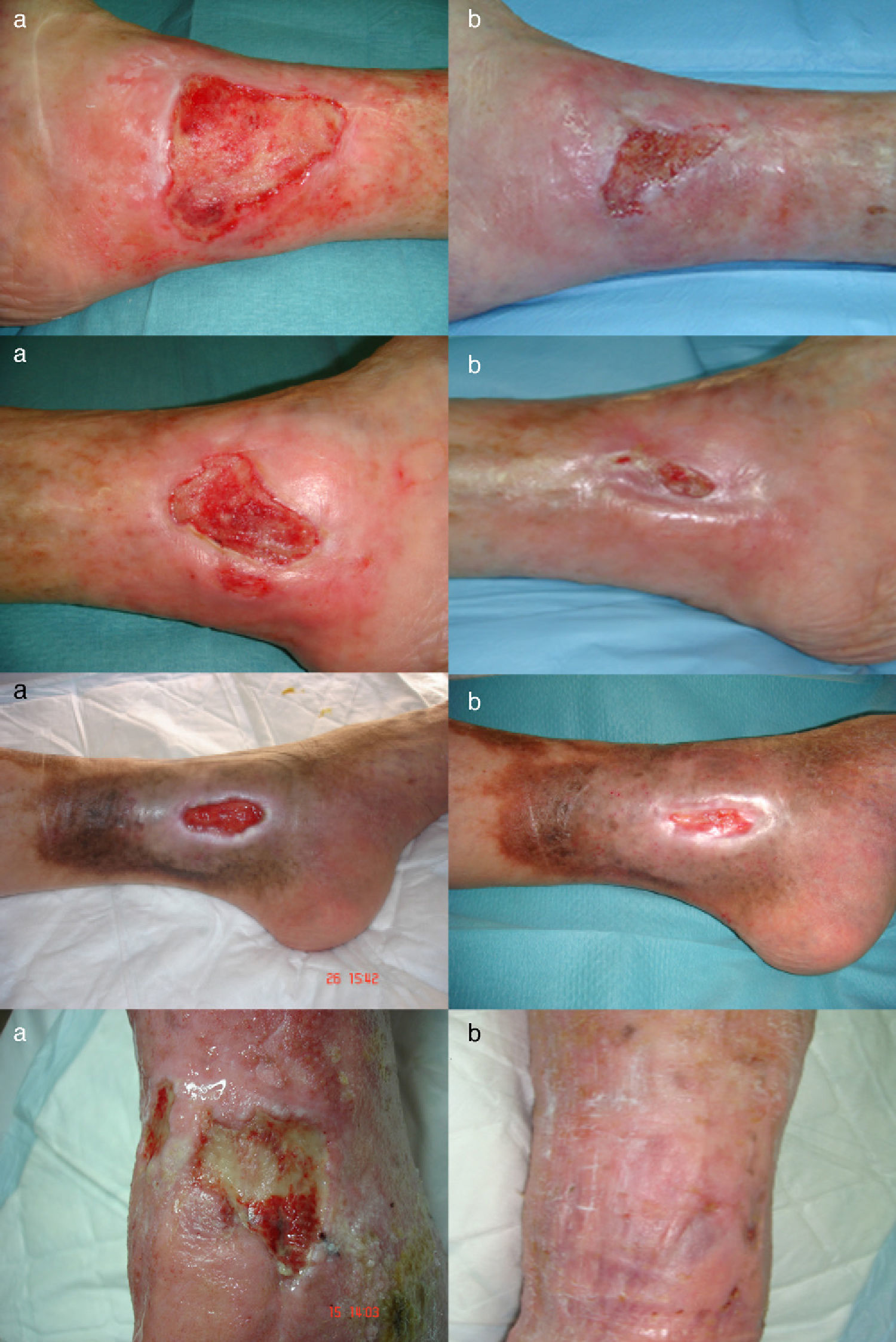
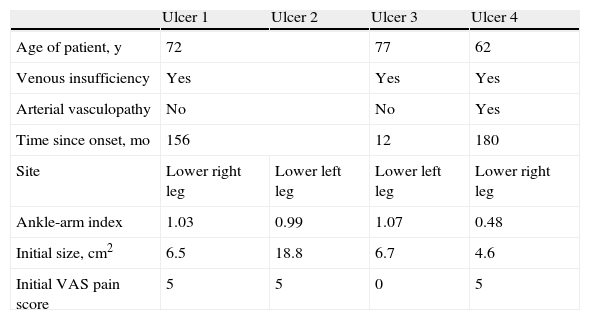
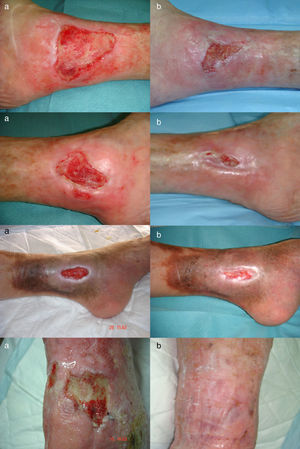
ResultsIn 3 patients we performed a single amniotic membrane transplantation on a total of 4 ulcers located on the distal third of the lower limbs, 2 on the left leg and 2 on the right leg. The mean age of the patients was 71.3 years. The ulcers were venous in 3 cases (ankle-arm index,>0.9) and arterial in 1 case (ankle-arm index, 0.48). The mean ulcer size was 9.15cm2 (range, 4.6-18.8cm2) and the mean time since onset was 126 months with a maximum of 180 months (Table 1).
Demographic and Clinical Data of Patients and Description of Ulcers.
| Ulcer 1 | Ulcer 2 | Ulcer 3 | Ulcer 4 | |
| Age of patient, y | 72 | 77 | 62 | |
| Venous insufficiency | Yes | Yes | Yes | |
| Arterial vasculopathy | No | No | Yes | |
| Time since onset, mo | 156 | 12 | 180 | |
| Site | Lower right leg | Lower left leg | Lower left leg | Lower right leg |
| Ankle-arm index | 1.03 | 0.99 | 1.07 | 0.48 |
| Initial size, cm2 | 6.5 | 18.8 | 6.7 | 4.6 |
| Initial VAS pain score | 5 | 5 | 0 | 5 |
Abbreviation: VAS, visual analog scale (0 being no pain and 10 the worst pain imaginable).
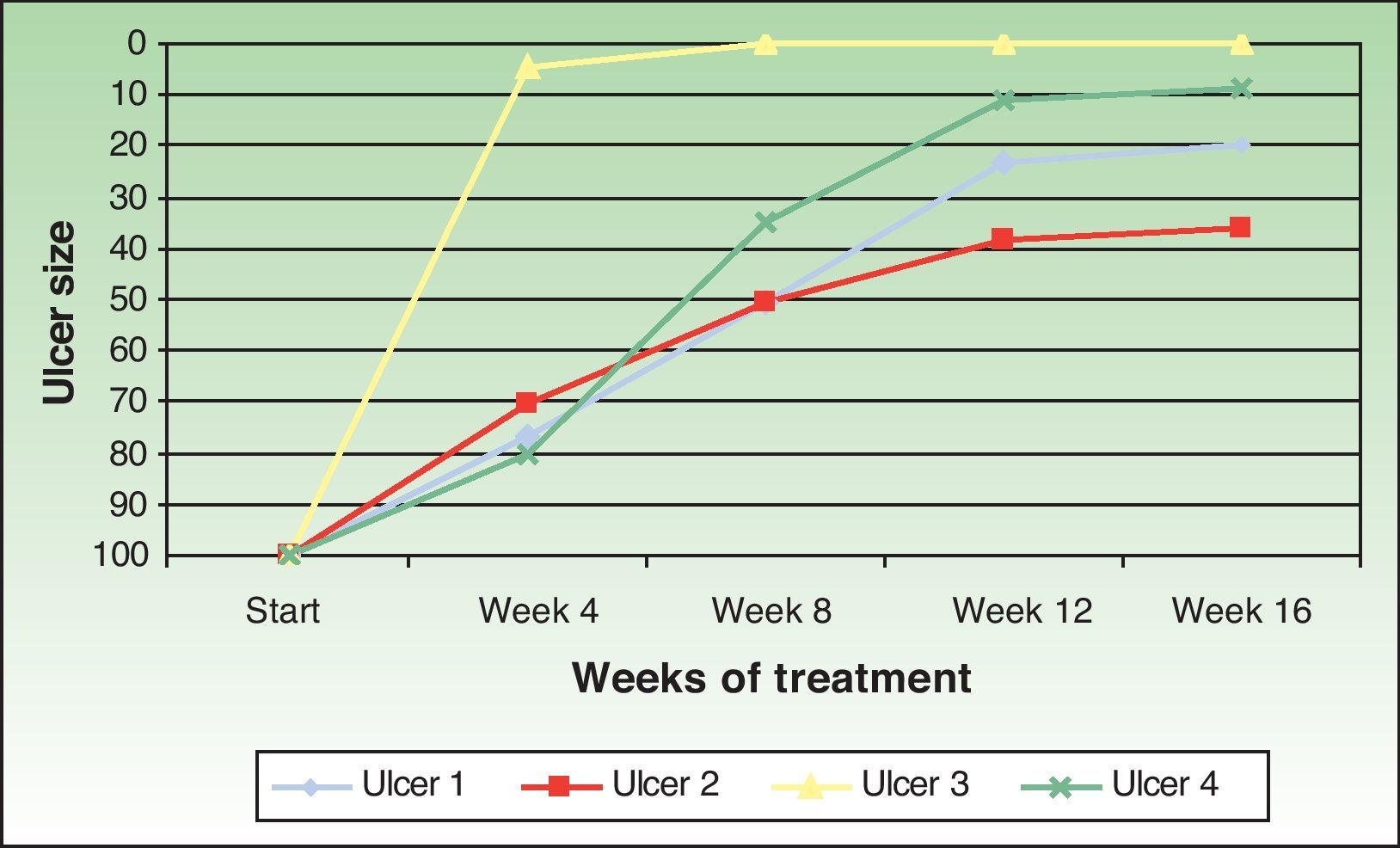
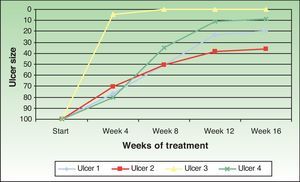
The ulcer size decreased progressively after the amniotic membrane transplantation (Fig. 1). In the follow-up visit in week 8 complete reepithelialization of 1 of the ulcers was observed. The mean reduction in ulcer size compared with baseline was 81.93% (range, 61.70%-100%) (Fig. 2)
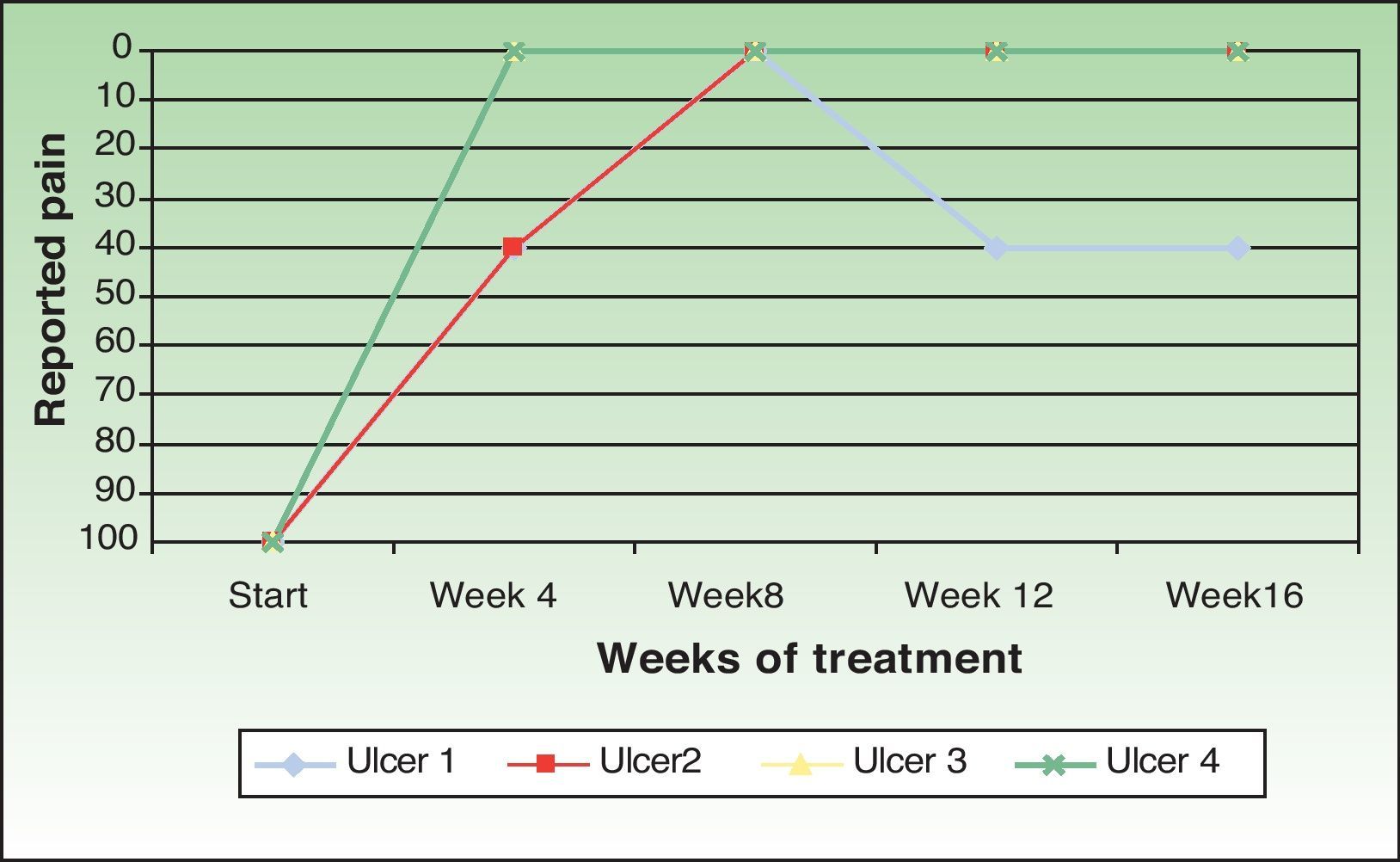
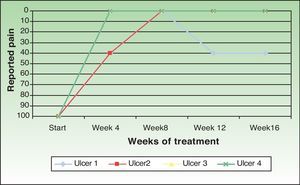
One patient (ulcer 3) reported no pain at any time during the study. Two patients reported the disappearance of pain perceived after the procedure. However, the patient with ulcer 2 reported an increase in pain in the follow-up visit at week 12 and this pain continued until the end of the study. Taking into account all cases the pain intensity decreased by 86.66% compared with baseline (Fig. 3).
Two of the patients had perilesional irritant dermatitis at week 4, so the foam dressing was replaced with a carbon and colloidal silver dressing and betamethasone and gentamicin cream was applied to the damaged skin as needed, with a follow-up visit after 7 days.
No clinical infection or increase in size was observed in any of the ulcers.
DiscussionThe use of amniotic membrane for treating skin lesions can be traced back to the early 20th century. Interest in their use in vascular ulcers of the lower limbs began to increase in the 1990s.7,8,11–13,16 This interest stems from the fact that the membrane's antimicrobial, antiinflammatory, analgesic, antiangiogenic, and reepithelializing properties5,7,9,10,12,17–21are considered ideal characteristics for a dressing.3 Amniotic membrane is also easy to obtain and does not require invasive application techniques.
This study is the first in Spain that has used amniotic membrane transplantation to foster or accelerate healing of refractory leg ulcers. It was a pilot study designed to test the advantages reported in the literature and therefore included only 4 cases.
The ulcers treated were mainly venous. The age of the patients, the time since onset, and the size of the ulcers were similar to those of other published cases. Complete reepithelialization was achieved in 1 of the 4 cases in approximately 6 weeks; a reduction in size of over 60% was achieved in the remaining ulcers at the end of the study period. These data are consistent with those obtained by other authors. Mermet et al.12 used amniotic membrane in 15 patients with mainly venous leg ulcers, observing after 3 months a reduction of not less than 50% in 12 patients (80%), of whom 3 (20%) achieved complete reepethelialization. Pesteil et al.8,13 used amniotic membrane in 8 patients with refractory arterial ulcers, observing complete reepithelialization in 2 of the patients (25%) after 26 weeks, a reduction of not less than 50% in a further 3 patients (about 35%) after 32 weeks, and no response in 3 patients.
In all cases amniotic membrane transplantation reduced pain significantly. This beneficial effect is also reported in the literature8,12,13; its basis is unknown, but it could result from the covering of the sensory nerve endings.20 The increase in pain intensity in 1 patient in our series suggests that it might be advisable to repeat the procedure in those cases in which pain increases.
No clinical infection was detected in any of the cases treated. This finding confirms that amniotic membrane can be safely grafted when it is harvested and handled following the strictest procedures. Its well-known antibacterial activity6,19may be due to several factors: presence of antibacterial substances in amniotic fluid, expression of antimicrobial peptides of the innate immune system, biological closure of the wound, or good adherence to the wound.19,20
One of the most valued features of amniotic membrane is its low immunogenicity22: no adverse reactions have been reported after its use. However, in the first few weeks of the study 2 of our patients developed irritant dermatitis, which responded to topical corticosteroids and a dressing change aimed at improving control of the exudate. We therefore considered that the dermatitis was not caused by a reaction to the membrane but by excess exudate.
Amniotic membrane is more expensive than common dressings. However, in a previous study comparing the cost-effectiveness of amniotic membrane with that of autografts and allografts of biocompatible skin substitutes for the treatment of vascular ulcers on the lower limbs, we concluded that amniotic membrane transplantation was the second most efficient option, after autografts.4
In conclusion, our results suggest that amniotic membrane may be an effective and safe alternative for treating refractory leg ulcers.
Conflict of InterestsDr. M. Alsina is currently director of the skin bank of the Transplant Services Foundation of Hospital Clínic de Barcelona but obtains no financial benefit from this post.
S. Pedregosa-Fauste reports no conflicts of interest.
Please cite this article as: Alsina-Gibert M, Pedregosa-Fauste S. Aplicación de membrane amniótica en el tratamiento de las úlceras crónicas de extremidades inferiores. Actas Dermosifiliogr. 2012;103:608–613.