Relapse rates in patients with acne after treatment with oral isotretinoin vary between 10% and 60% depending on the dosage regimen used, the length of follow-up, and the characteristics of the study population. The aim of this study was to determine the acne relapse rate within 2 years of completion of isotretinoin therapy and to identify the prognostic factors associated with relapse.
Material and methodsWe studied a series of patients with cystic acne who had received a cumulative therapeutic dose of at least 120mg/kg of oral isotretinoin. Data were analyzed using descriptive and analytical statistical methods. The relapse rate was expressed as the number of events divided by the amount of person-time. Factors predicting relapse were identified using multivariate Cox regression analysis.
ResultsA total of 142 patients were followed up for 24 months or until relapse occurred. The relapse rate was 15 events per 100 person-years of follow-up. The risk of relapse was twice as high among men. The protective effect of maintenance treatment with topical retinoids was 32% for each month of use. The risk of relapse was 3.5 times higher among women not receiving antiandrogen therapy.
ConclusionsThe relapse rate in our study was high and similar to that reported in the literature, Maintenance treatment with topical retinoids in men and women and androgen treatment in women helped to prevent relapse.
Las recaídas después del tratamiento con isotretinoína oral varían entre el 10 y el 60% según los esquemas utilizados, el tiempo de seguimiento y las características de la población estudiada. El objetivo de este estudio fue establecer la tasa de recaída durante los 2 primeros años de seguimiento postratamiento e identificar los factores pronóstico asociados con la recaída.
Material y métodoSe realizó un estudio de seguimiento a una cohorte de pacientes con acné quístico que completaron una dosis terapéutica total de mínimo 120mg/kg de isotretinoína oral. Se analizaron los datos utilizando herramientas de estadística descriptiva y analítica. Se calculó la tasa de recaída empleando el número de eventos sobre el denominador persona tiempo. Los factores pronóstico se establecieron a través del método de análisis multivariado de regresión de Cox.
ResultadosSe siguieron 142 pacientes durante 24 meses o hasta la ocurrencia del evento. La tasa de incidencia de recaída fue de 15 eventos por 100 personas/año de seguimiento. El riesgo de recaer fue el doble en los pacientes de sexo masculino. El tratamiento de mantenimiento con retinoides tópicos brindó una protección del 32% por cada mes de uso. Las mujeres que no tomaron tratamiento antiandrógeno tuvieron un riesgo de sufrir recaída 3,5 veces mayor.
ConclusionesLa tasa de recaída en nuestra población es alta y similar a la comunicada en otros estudios. El tratamiento de mantenimiento con retinoides tópicos en hombres y mujeres, y el tratamiento antiandrógeno en mujeres, ayudan a evitar recaídas.
Oral isotretinoin is the most effective treatment for cystic acne. Since its introduction over 30 years ago,1 the drug has become increasingly popular among patients and dermatologists worldwide.
Use of this agent quickly increased because of its effectiveness and the evidence provided by broad previous experience. Between 1992 and 2000, for example, prescription of oral isotretinoin increased 250% in the United States.2
Despite its effectiveness, isotretinoin should not be considered curative for cystic acne because new lesions often appear (relapses) after discontinuation of treatment. The relapse rates reported in different studies from around the world range from 10% to 60%, depending on the treatment regimens used and the length of follow-up (Table 1). Although predictive factors for relapse have been identified, the evidence is inconclusive and often contradictory.
Relapse Rates After Treatment With Oral Isotretinoin According to International Studies.
| Authors | Number of Patients | Relapse, % |
| Coloe J, et al.18 | 102 | 45 |
| Borghi A, et al.17 | 150 | 9 |
| Liu A, et al.23 | 405 | 23 |
| Azoulay L, et al.10 | 17351 | 41 |
| Quéreux G, et al.14 | 52 | 52 |
| Al-Mutairi N, et al.24 | 117 | 49 |
| Lehucher-Ceyrac D, et al.5 | 237 | 14 - 48 |
| White GM, et al.13 | 179 | 61 |
| Layton AM, et al.15 | 88 | 30 - 39 |
| Stainforth JM, et al.16 | 299 | 17 - 23 |
| Harms M, et al.4 | 86 | 15 |
| Jones DH, et al.20 | 76 | 55 |
In 1982, the first cases of relapse were reported in patients with acne following a course of treatment with high-dose oral isotretinoin (1mg/kg/d), with recovery of sebum production by the sebaceous glands at 16 weeks after discontinuation of treatment.3 Subsequently, the type of acne, the site of the lesions, age, male sex, and daily dose of isotretinoin were postulated as possible predictive factors for relapse, and the relapse rate was found to be greater with longer follow-up durations.3-5 From 2000 onwards, investigators in different countries started to publish studies pointing to higher relapse rates, and identified other risk factors for relapse, such as total therapeutic dose of isotretinoin, disease duration, family history of acne, and prior treatment with topical retinoids.6
The aim of this study was to establish the relapse rate during the first 2 years of follow-up after treatment with oral isotretinoin in patients with cystic acne and to attempt to identify predictive factors for relapse.
Materials and MethodsPatients with moderate or severe cystic acne who had been prescribed oral isotretinoin were studied. Disease severity was defined as moderate or severe according to the number of inflammatory lesions, the criterion normally used in our center (Table 2).7 The sample size was calculated for a 95% CI and a power of 80%. According to the calculation, 130 patients were required. Assuming 5% would be lost to follow-up, the protocol called for the inclusion of 137 patients.
The study enrolled patients resident in the urban area of Bogota DC, Columbia, who were included if they attended scheduled appointments during the treatment period, followed instructions, and discontinued treatment after completing a minimum total therapeutic dose of 120mg/kg. All patients gave informed consent.
Patients who met the inclusion criteria were recruited between July 31, 2006, and January 31, 2008. They were scheduled for appointments every 3 months from the moment they discontinued treatment with oral isotretinoin. At the first follow-up visit, the treating dermatologist prescribed maintenance therapy with topical retinoids (adapalene 0.1%, tretinoin 0.025%, or retinoic acid 0.025%); the initial regimen was 2 nights a week and the frequency of application was gradually increased depending on the patient's tolerance of the retinoid. Female patients were also prescribed oral contraception (cyproterone acetate 2.5mg and ethinylestradiol 0.035mg, chlormadinone acetate 2mg and ethynylestradiol 0.03mg), or spironolactone (100mg/d).
At each follow-up visit, the number of patients with relapse, as defined in Table 3, was compared with the number without recurrence to establish the relapse rate. The individual characteristics of those who relapsed and the differences between them and those who remained disease free were then analyzed according to the variables presented in Table 3.
Variables.
| Variable | Definition |
| Relapse | More than 15 open or closed comedones or a combination of both, or more than 15 papulopustular lesions, or 2 or more nodulocystic lesions |
| Sex | Male or female |
| Age | Years |
| Family history | First-degree relation |
| Acne severity | Mild, moderate, severe |
| History of prior treatment | With topical tretinoin |
| Daily dose | 0.5-1mg/kg |
| Total therapeutic dose | 120-150mg/kg |
| Maintenance therapy | Topical retinoids (men and women) |
| Oral contraceptives or spironolactone (women) |
The relapse rate was calculated as an incidence density (number of new relapses per person-time) to establish the period of individual follow-up and to gain a better understanding of the dynamic process driving the true relapse rate. The corresponding CIs were calculated.
For nominal variables, proportions and the Fisher exact test were used to establish whether or not an association was present. In the case of ordinal variables, proportions and medians were used when necessary. When continuous variables were distributed normally, the t test was used; otherwise, the log-rank nonparametric test was used. For the comparison between patients with and without relapse, measures of association were calculated (relative risk and hazard ratio) with their respective CIs and P values.
Each variable was analyzed in a bivariate model and a multivariate model using Cox regression. The level of significance for hypothesis testing was .05, that is, an association was considered significant when P≤.05.
The study was approved by the corresponding ethics committee. This was a minimum-risk study, in accordance with Resolution 8430/93 of the Ministry of Health and Social Services applicable in Columbia.8 The study was conducted in accordance with the tenets of the Declaration of Helsinki for clinical research in humans.9
ResultsThe study included 142 patients, who continued in follow-up until relapse or for 24 months after discontinuing treatment with oral isotretinoin, whichever occurred sooner. Seventy-nine of these patients (56%) were men and the mean (SD) age was 20 (4.7) years (range, 13-37 years). Fifty per cent of the sample was aged between 18 and 23 years.
Thirty-seven relapses were reported. Taking into account the individual follow-up duration for each subject, the total number of patient-years of follow-up was 242. Of the patients without relapse, 90% (94/105) had a follow-up of 24 months; the remaining 10% (11/105) completed follow-up periods of between 16 and 23 months. The group of 37 patients with relapse completed follow-up periods of between 3 and 24 months.
The relapse rate was 15 relapses per 100 person-years, that is, for every 100 person-years of follow-up, 15 new relapses would be expected (95% CI, 11-21 relapses).
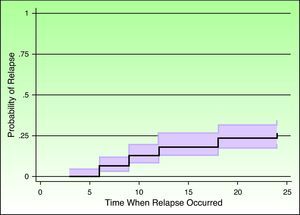
Survival was analyzed using the Kaplan-Meier method, which allowed the probability of experiencing an event to be calculated as a function of follow-up time. Table 4 shows the results of the survival analysis and Figure 1 plots the probability of relapse as a function of time.
Kaplan-Meier Survival Functions.
| Follow-up Period, mo | Patients in Follow-up, n | Event (Relapse) | Lost to Follow-up | Probability of No Relapse in This Period | Probability of Relapse in This Period | Probability of Relapse in This Period (95% CI) |
| 1-3 | 142 | 1 | 1 | .992 | .007 | .001-.049 |
| 4-6 | 140 | 8 | 0 | .936 | .063 | .033-.119 |
| 7-9 | 132 | 9 | 0 | .872 | .127 | .082-.194 |
| 10-12 | 123 | 8 | 0 | .815 | .184 | .129-.258 |
| 13-16 | 115 | 0 | 1 | .815 | .184 | .129-.258 |
| 17 | 114 | 0 | 1 | .815 | .184 | .129-.258 |
| 18 | 113 | 7 | 2 | .765 | .234 | .173-.314 |
| 19 | 104 | 0 | 1 | .765 | .234 | .173-.314 |
| 20-21 | 103 | 0 | 2 | .765 | .234 | .173-.314 |
| 22 | 101 | 0 | 2 | .765 | .234 | .173-.314 |
| 23 | 99 | 0 | 1 | .765 | .234 | .173-.314 |
| 24 | 98 | 4 | - | .733 | .266 | .200-.348 |
The mean age (21 years) was the same among patients who experienced a relapse and those who did not (P=.7 for difference of the means and P=.9 for difference of the medians).
Relapse Versus Daily DoseThe mean daily dose was 0.41mg/kg/d in patients with relapse and 0.39mg/kg/d in the group without relapse.
Relapse Versus Total Therapeutic Dose
The mean total therapeutic dose was 138mg/kg in those with relapse and 144mg/kg in the group without relapse.
Table 5 presents the bivariate analysis of the other variables studied.
Risk Factors for Relapse: Bivariate Analysis.
| Variable | Total, n | Relapse, n | Relapse, % | Relative Risk | 95% CI | P |
| Male sex | 79 | 26 | 32 | 1.8 | 1.01-3.50 | .037 |
| Family history | 109 | 30 | 27 | 1.29 | 0.62-2.67 | .46 |
| Severe acne | 98 | 26 | 26 | 0.94 | 0.51-1.73 | .84 |
| Prior topical tretinoin | 14 | 6 | 42 | 1.7 | 0.89-3.48 | .13 |
| Daily dose <0.5mg/kg | 111 | 30 | 27 | 1.2 | 0.6-2.4 | .6 |
| Topical treatment (maintenance) | 131 | 26 | 19 | 0.19 | 0.14-0.27 | .001 |
| Antiandrogen (maintenance) | 57 | 8 | 14 | 0.28 | 0.1-0.78 | .027 |
In the model constructed to predict relapses, significant results were obtained for sex (hazard ratio, 2.3; 95% CI, 1.02-5.2; P = .043) and topical maintenance treatment (hazard ratio, 0.68; 95% CI, 0.6-0.77; P = .001) (Table 6).
DiscussionAfter publication of the first studies reporting relapse following the treatment of acne with oral isotretinoin,3 it was debated whether the factors that favored relapse were associated with the individual patient or the treatment regimen. To date, the information available is contradictory, possibly because the studies with data on relapse are not comparable due to large differences in inclusion criteria, treatment regimen, definition of relapse, and follow-up duration.
The cohort in this study was a dynamic one, that is, patients entered and left the study at different time-points. We calculated that 15 relapses could be expected for every 100 patient-years of follow-up, a rate similar to some of those reported in the literature (Table 1).
In our study, 32% of men (26/79) experienced a relapse compared to 17% of women (11/63). Male sex was a prognostic factor for relapse, as reported in previous studies,4,5,10 perhaps because of the antiandrogen treatment prescribed for women in this cohort. The presence of hyperandrogenism, like other endocrine disorders,11 has also been associated with therapeutic failure of oral isotretinoin.12
The patient's age at the start of therapy with oral isotretinoin has been reported to be a predictive factor for relapse, with a greater risk found in patients starting at under 15,4 16,10 18,13 or 20 years.14 In our study, age was not a statistically significant predictive factor.
Quéreux et al.14 concluded that family history of acne was a predictive factor for relapse. In our cohort, we found no significant positive or negative association between relapse and family history of acne (Table 5).
Acne severity is another predictive factor often described.5,15,16 However, in our study, we found no statistical or epidemiological association between severity and relapse. A possible bias in the analysis of this variable is that we did not use a validated scale to classify disease severity.
A history of prior treatment with topical tretinoin, suggested by Quéreux et al.14 as a predictive factor for relapse, may be related to confounding variables such as disease severity, as it is likely that those with severe acne have a history of prior consultation and prescription of topical retinoids before initiating treatment with oral isotretinoin.
Topical retinoid maintenance treatment was the most important protective factor in our study. A recent study that took into account maintenance therapy with topical retinoids reported a very low relapse rate (9%) during the first year after discontinuation of oral isotretinoin even though low doses of that drug had been used.17 According to our results, the relapse rate was 19% (26/131) in patients who continued with topical maintenance therapy with retinoids but 100% (11/11) in those who discontinued the maintenance regimen. Thus, maintenance therapy with a topical retinoid was a protective factor. For every additional month of topical maintenance therapy, the probability of avoiding relapse was 32% lower (95% CI, 23%-40%). Thus, regular use of topical retinoids during the first 2 years after discontinuing treatment with oral isotretinoin was a protective factor against relapse, with statistically significant results (Table 6).
Maintenance treatment with oral contraceptives or spironolactone also helps to prevent relapse in women. It has been reported that failure to control hormonal imbalances in women with acne may be a predisposing factor to therapeutic failure during treatment with oral isotretinoin5; however, to date, antiandrogen therapy has not been considered a protective factor. In our study, the relative risk of relapse was 0.28 among women who took antiandrogen therapy after finishing treatment with oral isotretinoin compared to those who did not; thus, women who did not take any such therapy had a risk 3.5 times greater of experiencing relapse. This finding suggests that taking antiandrogen maintenance therapy is a protective factor for relapse in women, with statistically significant results.
In 1993, Layton et al.15 established that taking suboptimal doses, less than 120mg/kg, is a predictive factor for relapse. Patients who did not take this minimum dose were excluded from our study. However, no statistically significant difference was observed between the mean total therapeutic dose taken by the patients who relapsed and those who did not. Coloe et al18 also found that the total therapeutic dose of isotretinoin bore no relationship to relapse 1 year after finishing treatment.
An association between relapse and low daily doses of oral isotretinoin was suggested by White et al.,13 in patients who received less than 1mg/kg/d, and by Layton et al.,15 Leyden,19 and Jones and Cunliffe20 in patients who received less than 0.5mg/kg/d. Although the average daily dose taken by patients in our study was 0.41mg/kg/d, the relapse rate among those who received treatment with doses lower than 0.5mg/kg/d was similar to that of patients with received doses above 0.5mg/kg/d (Table 5). We did not find an association between taking a dose less than 0.5mg/kg/d and relapse, in agreement with 3 recent studies in which the relapse rate was similar in patients who took high doses (>0.5mg/kg/d) and low doses (0.25–0.4mg/kg/d).17,21,22
In conclusion, the incidence of relapse in patients with cystic acne after the first cycle of treatment with oral isotretinoin is 15 events per 100 person-years of follow-up. This incidence is considered high for our population and similar to that reported in other studies. Follow-up can be extended in the 142 patients in our study beyond 2 years to enable comparison of the current results with those obtained at 5 years or later.
Of all the variables analyzed in our study, only male sex could be considered a clear predictive factor for relapse. These findings suggest that there may be individual susceptibility that we are as yet unaware of that determines response to treatment and predisposition to relapse, independent of the variables we studied.
Maintenance therapy with a topical retinoid, applied regularly for the first 2 years after completing treatment with oral isotretinoin, and oral antiandrogen treatment in women were 2 key protective factors for decreasing the relapse rate.
Knowledge of the factors described above could be the first step towards designing new strategies to reduce the risk of relapse during follow-up of these patients.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
We would like to thank Dr. María Cristina Ronderos-Acevedo for her contribution to the initial design of the study and Dr. Luisa Porras de Quintana for her support in preparing the project and reviewing the study protocol.
Please cite this article as: Morales-Cardona CA, Sánchez-Vanegas G. Tasa de recaída y factores pronóstico de recaída después del tratamiento con isotretinoína oral en pacientes con acné quístico. Actas Dermosifiliogr.2013;104:61-6.