Hyperhidrosis, or excessive sweating, is characterized by overactivity of the eccrine sweat glands, usually associated with dysfunction of the autonomic nervous system. Primary focal hyperhidrosis is the most common form and can affect the axillae, palms, soles, and/or face, often leading to significantly impaired quality of life and social functioning. Treatment is complex. Topical antiperspirants are normally recommended as the first-line treatment for mild hyperhidrosis. Multiple clinical trials and prospective studies support the efficacy and tolerability of oral and topical anticholinergics in the management of hyperhidrosis. Topical glycopyrronium, which has been investigated in at least 8 clinical trials enrolling more than 2000 patients, is probably the first-line pharmacological treatment for axillary hyperhidrosis in patients with moderate to severe disease poorly controlled with topical antiperspirants. Second-line treatments include botulinum toxin injections, microwave treatment, and oral anticholinergics. We review the use of topical anticholinergics in the management of focal hyperhidrosis in adults and children.
La hiperhidrosis se caracteriza por excesiva sudoración, habitualmente secundaria a disfunción autonómica con hipersecreción de las glándulas sudoríparas ecrinas. La hiperhidrosis primaria focal es la forma más frecuente, y afecta axilas, palmas, plantas y/o cara. Frecuentemente genera un gran impacto en la calidad de vida y en la actividad social. Su tratamiento es complejo. Los antitranspirantes tópicos son recomendados en primer lugar en la mayoría de casos de hiperhidrosis leve. Múltiples ensayos clínicos y estudios prospectivos avalan la eficacia y tolerabilidad de los anticolinérgicos orales y tópicos. En casos moderado/graves, el glicopirronio tópico, el cual ha sido evaluado en al menos 8 ensayos clínicos con más de 2.000 pacientes en total, podría ser considerado la primera línea farmacológica en la hiperhidrosis axilar mal controlada con antitranspirantes tópicos; seguido por inyecciones de toxina botulínica, sistemas de microondas y por anticolinérgicos orales. En este artículo revisamos el rol de los anticolinérgicos tópicos en el manejo de la hiperhidrosis focal en adultos y niños.
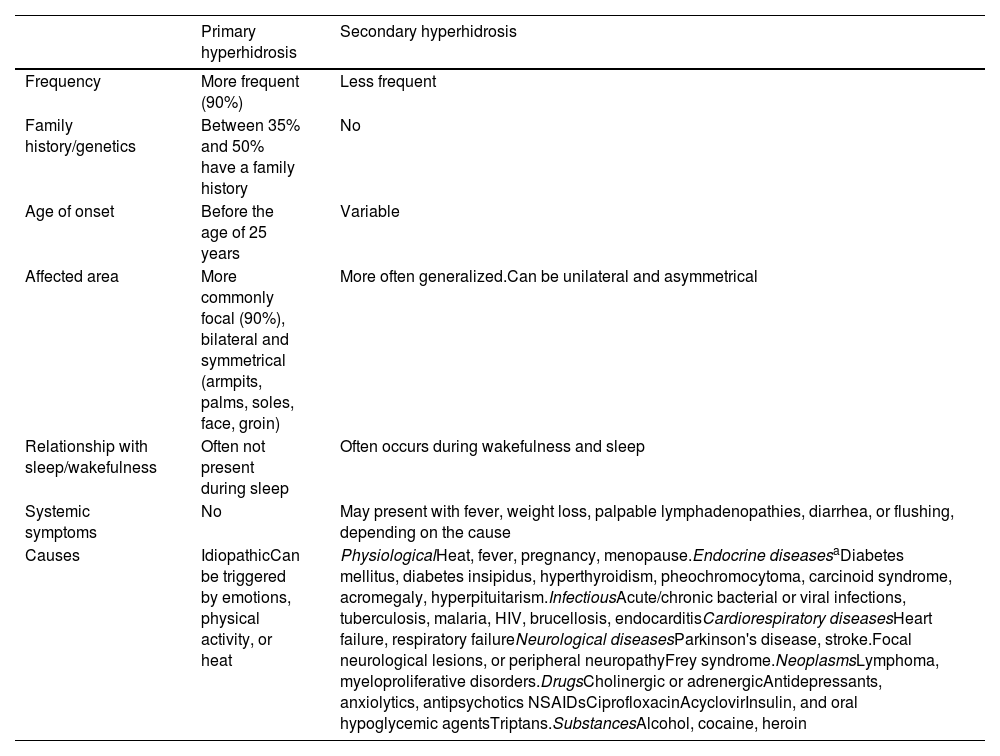
Hyperhidrosis is characterized by excessive sweating, and is often due to autonomic dysfunction inducing the hypersecretion of eccrine sweat glands.1 It affects nearly 5% of the population,2 and usually starts between the ages of 14 and 25.1,2 Hyperhidrosis can be categorized as primary (in over 90% of the cases), or secondary. Primary hyperhidrosis (PH) is associated with excessive neurogenic activity on normal eccrine glands. Its pathophysiology is complex and can be due to a dysfunctional autonomic nervous system, and/or abnormal emotional control.3 Secondary hyperhidrosis is induced by various disorders such as infections, neoplasms, neuroendocrine disorders, or drugs (Table 1).1
Primary and secondary hyperhidrosis.
| Primary hyperhidrosis | Secondary hyperhidrosis | |
|---|---|---|
| Frequency | More frequent (90%) | Less frequent |
| Family history/genetics | Between 35% and 50% have a family history | No |
| Age of onset | Before the age of 25 years | Variable |
| Affected area | More commonly focal (90%), bilateral and symmetrical (armpits, palms, soles, face, groin) | More often generalized.Can be unilateral and asymmetrical |
| Relationship with sleep/wakefulness | Often not present during sleep | Often occurs during wakefulness and sleep |
| Systemic symptoms | No | May present with fever, weight loss, palpable lymphadenopathies, diarrhea, or flushing, depending on the cause |
| Causes | IdiopathicCan be triggered by emotions, physical activity, or heat | PhysiologicalHeat, fever, pregnancy, menopause.Endocrine diseasesaDiabetes mellitus, diabetes insipidus, hyperthyroidism, pheochromocytoma, carcinoid syndrome, acromegaly, hyperpituitarism.InfectiousAcute/chronic bacterial or viral infections, tuberculosis, malaria, HIV, brucellosis, endocarditisCardiorespiratory diseasesHeart failure, respiratory failureNeurological diseasesParkinson's disease, stroke.Focal neurological lesions, or peripheral neuropathyFrey syndrome.NeoplasmsLymphoma, myeloproliferative disorders.DrugsCholinergic or adrenergicAntidepressants, anxiolytics, antipsychotics NSAIDsCiprofloxacinAcyclovirInsulin, and oral hypoglycemic agentsTriptans.SubstancesAlcohol, cocaine, heroin |
NSAID, nonsteroidal anti-inflammatory drugs.
Focal hyperhidrosis (FH) is the most common form of PH and typically affects the armpits, palms, soles, groin, inframammary fold, and/or craniofacial area.1 It can trigger skin maceration, fungal or bacterial infections, and irritative dermatitis. FH can have a significant impact on the patients’ quality of life, affecting social activities, exercise, work, and studies.4–6 A recent systematic review found rates of anxiety in up to 49% of these patients, and rates of depression in up to 60% of them.5 In Spain, a cross-sectional study (n = 100) described a 20% rate of depression and a proportional relationship between the severity of hyperhidrosis and depressive symptoms.4 Hyperhidrosis runs underdiagnosed and undertreated in children, and can impact the quality of life significantly, even in children younger than 12 years.7
The management of FH is complex. There are multiple topical, systemic, minimally invasive, and surgical alternatives currently available. Topical antiperspirants are generally recommended as a first-line therapy, especially to treat cases of mild FH.8 In cases of moderate-to-severe FH that remain unresponsive to topical antiperspirants, topical anticholinergics,8 or oral anticholinergics,9,10 botulinum toxin injections,11,12 and various devices such as iontophoresis, radiofrequency,13 ultrasound, and microwave thermolysis14 are often used. In refractory cases, various surgical techniques such as sympathectomy can be useful.15 In this article, wéll be reviewing the role of topical anticholinergics in the management of HF in both children and adults.
Clinical features of focal hyperhidrosis in children and adultsIn prepubertal individuals, FH primarily affects the palms and soles in 90% of the cases followed by the armpits and the face (15% and 6%, respectively).1,16 In adults, the most common location is the armpits (50%), followed by the soles of the feet (30%), palms (24%), and face (10%).1 Simultaneous involvement of multiple regions of interest is a common finding.1 FH may have a lower prevalence in the elderly, suggesting regression of the condition in advanced age.1
DiagnosisIn most cases, the diagnosis is clinical. However, in patients in whom hyperhidrosis appears past the age of 25 with generalized, asymmetrical, or unilateral hyperhidrosis, or during sleep, or with systemic symptoms (Table 1) secondary hyperhidrosis should be ruled out, and the corresponding additional tests should be performed based on clinical suspicion. The most common secondary causes are endocrine disorders such as diabetes mellitus, hyperthyroidism, and hyperpituitarism, followed by neurological disorders.17
Among the assessment scales used in the management of hyperhidrosis, the most widely used ones, according to a recent systematic review, are the Hyperhidrosis Disease Severity Scale (HDSS) (Table 2), and the Dermatology Life Quality Index (DLQI). A different scale used is the Hyperhidrosis Quality of Life Index (HidroQoL).6 An objective method of assessment is to measure sweat production by gravimetry.
Hyperhidrosis Disease Severity Scale (HDSS).
| How would you categorize the intensity of your hyperhidrosis? | |
|---|---|
| Scorea | Response |
| 1 | My sweating is never noticeable and never interferes with my daily activities. |
| 2b | My sweating is tolerable but occasionally interferes with my daily activities. |
| 3b | My sweating is barely tolerable and often interferes with my daily activities. |
| 4b | My sweating is intolerable and always interferes with my daily activities. |
The goals of hyperhidrosis treatment are to reduce sweating and improve the patients’ quality of life. This can be achieved by reducing sweat production by the sweat glands, and/or the number of these.11 Some basic recommendations18 are summarized in Table 3.
Recommendations for patients with hyperhidrosis.
| Avoid tight clothing, synthetic garments, and occlusive footwear |
| Use drying powder (talc) |
| Use an additional layer of clothing to conceal sweat |
| Avoid spicy foods and alcohol |
| Avoid environments or situations that trigger emotional responses worsening the condition |
| For plantar hyperhidrosis, change socks and shoes frequently and use absorbent insoles |
Overall, topical antiperspirants are recommended as the first-line therapy to treat axillary and palmoplantar FH.11,18 However, they tend to be more effective in milder forms.11 Antiperspirants are over-the-counter topical products that contain mineral salts, such as aluminum salts, in various concentrations (6%,19 15%,20 20%,21,22 or 30%,23 among others). Their adverse events (AEs) include local irritation.18 Table 4 explains the mechanism of action and recommendations for their use. Despite being widely used in the routine clinical practice,24 we have not found any studies conducted in children to date on the use and tolerability of antiperspirant products (aluminum salts).
Concentration, mechanism of action, and usage recommendations for antiperspirants in the management of focal hyperhidrosis.
| Concentration | Over-the-counter antiperspirants (stick or roll-on) contain a maximum concentration of 12.5% of ACH.Other compounds and formulas can contain up to 40% ACH.Less commonly used salts include zirconium, vanadium, and indium. |
| Vehicles | Antiperspirants can be found in water, alcohol, glycerol (stick or roll-on), creams, or lotions.Lotions may allow for a more uniform application of the product. |
| Mechanism of action | Aluminum salts interact with mucopolysaccharides and form precipitates that block the lumen of eccrine sweat glands, but sweat production persists.It may induce atrophy of eccrine glands. |
| Recommendations | They should be applied to dry skin before bedtime.They should be left on for 8hours before washing the area.Apply daily for the first few weeks until a reduction in sweating is observed, then down-titrate to once or twice a week. |
| Adverse events | Appearance of miliaria in the applied area (due to sweat retention).Irritative dermatitis, dependent on ACH concentration.No association has been demonstrated with Alzheimer's disease, or breast carcinoma. |
Anticholinergics reduce sweating by competitively blocking the muscarinic receptors of acetylcholine in the eccrine sweat glands.
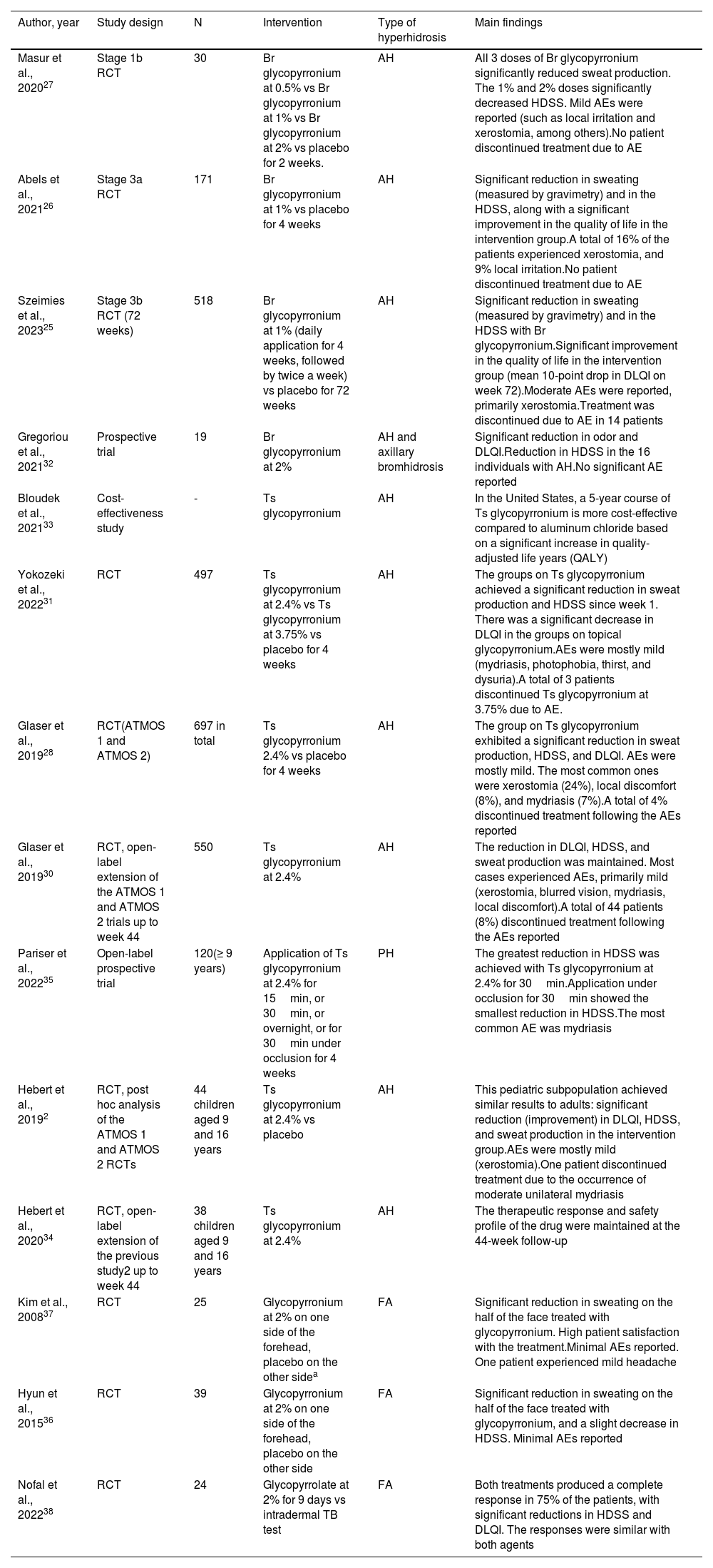
Topical glycopyrroniumAxillary hyperhidrosisTopical glycopyrronium has been approved by the Food and Drug Administration (FDA) to treat axillary hyperhidrosis (AH) in patients ≥ 9 years old. A recent phase-3b randomized clinical trial (RCT) evaluated the long-term effectiveness (72 weeks) of 1% glycopyrronium bromide in 518 patients with AH. It demonstrated a significant reduction in sweating (measured by gravimetry) and HDSS, as well as a significant improvement in the quality of life of the intervention group, with a decrease in HidroQoL and a mean reduction of 10 points in DLQI on week 72. The treatment was well-tolerated, and most AEs were mild (mainly xerostomia). It was discontinued due to the AEs reported in 14 cases.25 Similar results were seen in 2 previous phase-3a,26 and phase-1b27 RCTs of 171 and 30 patients, respectively (Table 5).
Studies on topical glycopyrronium for the management of focal hyperhidrosis.
| Author, year | Study design | N | Intervention | Type of hyperhidrosis | Main findings |
|---|---|---|---|---|---|
| Masur et al., 202027 | Stage 1b RCT | 30 | Br glycopyrronium at 0.5% vs Br glycopyrronium at 1% vs Br glycopyrronium at 2% vs placebo for 2 weeks. | AH | All 3 doses of Br glycopyrronium significantly reduced sweat production. The 1% and 2% doses significantly decreased HDSS. Mild AEs were reported (such as local irritation and xerostomia, among others).No patient discontinued treatment due to AE |
| Abels et al., 202126 | Stage 3a RCT | 171 | Br glycopyrronium at 1% vs placebo for 4 weeks | AH | Significant reduction in sweating (measured by gravimetry) and in the HDSS, along with a significant improvement in the quality of life in the intervention group.A total of 16% of the patients experienced xerostomia, and 9% local irritation.No patient discontinued treatment due to AE |
| Szeimies et al., 202325 | Stage 3b RCT (72 weeks) | 518 | Br glycopyrronium at 1% (daily application for 4 weeks, followed by twice a week) vs placebo for 72 weeks | AH | Significant reduction in sweating (measured by gravimetry) and in the HDSS with Br glycopyrronium.Significant improvement in the quality of life in the intervention group (mean 10-point drop in DLQI on week 72).Moderate AEs were reported, primarily xerostomia.Treatment was discontinued due to AE in 14 patients |
| Gregoriou et al., 202132 | Prospective trial | 19 | Br glycopyrronium at 2% | AH and axillary bromhidrosis | Significant reduction in odor and DLQI.Reduction in HDSS in the 16 individuals with AH.No significant AE reported |
| Bloudek et al., 202133 | Cost-effectiveness study | - | Ts glycopyrronium | AH | In the United States, a 5-year course of Ts glycopyrronium is more cost-effective compared to aluminum chloride based on a significant increase in quality-adjusted life years (QALY) |
| Yokozeki et al., 202231 | RCT | 497 | Ts glycopyrronium at 2.4% vs Ts glycopyrronium at 3.75% vs placebo for 4 weeks | AH | The groups on Ts glycopyrronium achieved a significant reduction in sweat production and HDSS since week 1. There was a significant decrease in DLQI in the groups on topical glycopyrronium.AEs were mostly mild (mydriasis, photophobia, thirst, and dysuria).A total of 3 patients discontinued Ts glycopyrronium at 3.75% due to AE. |
| Glaser et al., 201928 | RCT(ATMOS 1 and ATMOS 2) | 697 in total | Ts glycopyrronium 2.4% vs placebo for 4 weeks | AH | The group on Ts glycopyrronium exhibited a significant reduction in sweat production, HDSS, and DLQI. AEs were mostly mild. The most common ones were xerostomia (24%), local discomfort (8%), and mydriasis (7%).A total of 4% discontinued treatment following the AEs reported |
| Glaser et al., 201930 | RCT, open-label extension of the ATMOS 1 and ATMOS 2 trials up to week 44 | 550 | Ts glycopyrronium at 2.4% | AH | The reduction in DLQI, HDSS, and sweat production was maintained. Most cases experienced AEs, primarily mild (xerostomia, blurred vision, mydriasis, local discomfort).A total of 44 patients (8%) discontinued treatment following the AEs reported |
| Pariser et al., 202235 | Open-label prospective trial | 120(≥ 9 years) | Application of Ts glycopyrronium at 2.4% for 15min, or 30min, or overnight, or for 30min under occlusion for 4 weeks | PH | The greatest reduction in HDSS was achieved with Ts glycopyrronium at 2.4% for 30min.Application under occlusion for 30min showed the smallest reduction in HDSS.The most common AE was mydriasis |
| Hebert et al., 20192 | RCT, post hoc analysis of the ATMOS 1 and ATMOS 2 RCTs | 44 children aged 9 and 16 years | Ts glycopyrronium at 2.4% vs placebo | AH | This pediatric subpopulation achieved similar results to adults: significant reduction (improvement) in DLQI, HDSS, and sweat production in the intervention group.AEs were mostly mild (xerostomia).One patient discontinued treatment due to the occurrence of moderate unilateral mydriasis |
| Hebert et al., 202034 | RCT, open-label extension of the previous study2 up to week 44 | 38 children aged 9 and 16 years | Ts glycopyrronium at 2.4% | AH | The therapeutic response and safety profile of the drug were maintained at the 44-week follow-up |
| Kim et al., 200837 | RCT | 25 | Glycopyrronium at 2% on one side of the forehead, placebo on the other sidea | FA | Significant reduction in sweating on the half of the face treated with glycopyrronium. High patient satisfaction with the treatment.Minimal AEs reported. One patient experienced mild headache |
| Hyun et al., 201536 | RCT | 39 | Glycopyrronium at 2% on one side of the forehead, placebo on the other side | FA | Significant reduction in sweating on the half of the face treated with glycopyrronium, and a slight decrease in HDSS. Minimal AEs reported |
| Nofal et al., 202238 | RCT | 24 | Glycopyrrolate at 2% for 9 days vs intradermal TB test | FA | Both treatments produced a complete response in 75% of the patients, with significant reductions in HDSS and DLQI. The responses were similar with both agents |
AE, adverse events; AH, axillary hyperhidrosis; Br, bromide; DLQI, Dermatology Life Quality Index; FA, facial hyperhidrosis; HDSS, Hyperhidrosis Disease Severity Scale; min, minutes; PH, palmar hyperhidrosis; RCT, randomized clinical trial; Ts, tosylate; vs, versus.
In 2 RCTs (697 patients with AH),28,29 a 4-week course of 2.4% glycopyrronium tosylate induced a significant reduction in sweat production, HDSS, and DLQI. AEs were mostly mild [xerostomia (24%), local discomfort (8%), and mydriasis (7%)]. Four percent discontinued treatment due to the AEs reported. The duration of these RCTs (44 weeks) confirmed the maintained response and safety profile of the drug.30 Another Japanese RCT (n = 497) comparing 2.4% vs 3.75% tosylate glycopyrronium vs placebo showed similar results with significant clinical improvement in both intervention groups.31
In a prospective study of 19 patients with axillary bromhidrosis, the application of 2% topical glycopyrronium reduced odor and DLQI significantly, as well as HDSS in the 16 individuals with AH.32
Finally, a cost-effectiveness analysis conducted in the United States revealed that topical glycopyrronium was cost-effective in the management of AH compared to topical antiperspirants with aluminum chloride.33
In children, a post hoc analysis of 2 RCTs, including 44 individuals aged 9 to 16 years with AH, demonstrated that they achieved similar results compared to adults, with a significant reduction (improvement) of DLQI, HDSS, and sweat production using 2.4% tosylate glycopyrronium. AEs were mild and transient, and only in 1 case they led to drug discontinuation (transient moderate unilateral mydriasis).2 In the 44-week study extension period of this RCT of 38 children, the efficacy and safety profile of the drug was maintained.34 Children are adviced to not touch the cream with their hands, as they could later touch their eyes with it, causing mydriasis.24
Palmar hyperhidrosisA prospective study (n = 120) showed that tosylate glycopyrronium at various concentrations could be a good option to treat PH.35
Facial hyperhidrosisA RCT of 39 patients with FH demonstrated a significant reduction in sweating on the face side treated with the anticholinergic drug, along with a slight decrease in the HDSS.36 Similar results were seen in a previous RCT of 25 participants.37 In a recent RCT of 24 participants, both 2% topical glycopyrronium and botulinum toxin (BTX) injections achieved similar results, with a complete response in 75% of individuals with FH.38
Topical oxybutyninA RCT of 61 patients with FH in different locations (axillae, palms, or soles) demonstrated that the administration of 10% topical oxybutynin reduced both the DLQI and the HDSS significantly.39 Another recent RCT of 30 patients with PH studied the administration of 1% oxybutynin gel vs 1% oxybutynin nanoemulgel and described satisfactory results for both formulations.40 In a large phase 3 RCT (n = 244), a 20% oxybutynin lotion reduced sweat volume in patients with PH. Fifty-three per cent of the intervention group experienced less sweating by, at least, 50%, and 1 patient achieved a significant improvement in DLQI. No serious AEs were reported.41
In children, a pilot study (n = 10) with 3% topical oxybutynin included 4 adolescents aged 13 to 16 years with AH. Three completed the study and achieved a significant reduction in axillary sweating. The drug was well-tolerated.42
Topical sofpironium bromideSofpironium bromide has been approved in Japan to treat AH in adults. A phase III RCT (n = 281) demonstrated that a 6-week course of 5% sofpironium bromide induced a significant reduction in HDSS, and sweating measured by gravimetry in patients with AH. AEs were mostly mild and reported in 20% of individuals. Only 1 individual discontinued treatment due to the presence of AEs.43 The efficacy and safety profile were maintained in the 52-week study extension period.44 A recent prospective study (n = 80) demonstrated that a reduction in HDSS could be observed as early as in the 1st week of treatment.31
Topical umeclidiniumA phase 2a RCT (n = 23) revealed that a 2-week course of 1.85% umeclidinium significantly reduced axillary sweating and HDSS in over 40% of the patients with AH.45
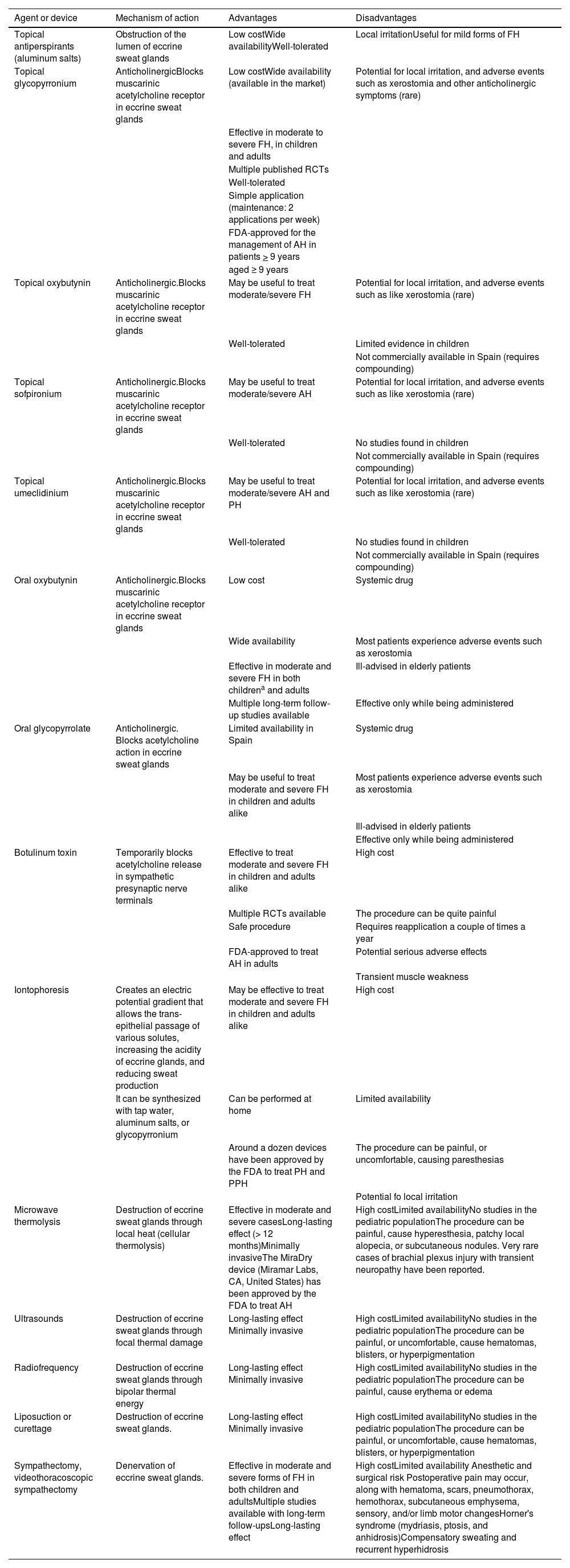
DiscussionThe management of hyperhidrosis can be complex, with multiple therapeutic alternatives available, including pharmacological treatments, medical devices, or surgical procedures. Table 6 shows the advantages and disadvantages of these therapies. Currently, the FDA-approved pharmacological therapies to treat AH include topical glycopyrronium and botulinum toxin A (BTX-A).15,18 No other drugs have ever been approved by the FDA for the management of FH in different parts of the body. Systemic anticholinergics such as oral oxybutynin are widely used off-label in the routine clinical practice, both in children and adults, with multiple studies showing good results to treat FH in general (axillary, palmoplantar, and craniofacial) with acceptable tolerability.10,15,46–51 Multiple RCTs, prospective studies, and systematic reviews support the effectiveness and tolerance of topical anticholinergics to treat FH, including AH, PH, PPH, and CFH.11,24,42,52. Topical glycopyrronium, which has been evaluated in, at least, 8 RCTs (including nearly 2000 patients)25–28,31,36,37is the topical drug on which more evidence has been accumulated to this date. Its use may limit the development of anticholinergic symptoms such as xerostomia seen in nearly 12% of the patients on long-term topical glycopyrronium therap.25 However, it is present in around 75% of the patients treated with systemic anticholinergics such as oxybutynin.10 It can also reduce serious AEs, and the risk of therapy discontinuation, reported in 2.7% of the cases on long-term topical glycopyrronium, and 17% of the patients on oral oxybutynin.49 We should mention that in recent years, several studies have suggested that there is an association between the use of systemic anticholinergics and the development of dementia, and caution is advised when prescribing them to middle-aged or elderly patients.53,54 Topical glycopyrronium has a level of evidence 1+, and a grade A recommendation for the management of AH, according to the Scottish Intercollegiate Guidelines Network (SIGN) classification.
Therapeutics available for the management of focal hyperhidrosis.
| Agent or device | Mechanism of action | Advantages | Disadvantages |
|---|---|---|---|
| Topical antiperspirants (aluminum salts) | Obstruction of the lumen of eccrine sweat glands | Low costWide availabilityWell-tolerated | Local irritationUseful for mild forms of FH |
| Topical glycopyrronium | AnticholinergicBlocks muscarinic acetylcholine receptor in eccrine sweat glands | Low costWide availability (available in the market) | Potential for local irritation, and adverse events such as xerostomia and other anticholinergic symptoms (rare) |
| Effective in moderate to severe FH, in children and adults | |||
| Multiple published RCTs | |||
| Well-tolerated | |||
| Simple application (maintenance: 2 applications per week) | |||
| FDA-approved for the management of AH in patients > 9 years | |||
| aged ≥ 9 years | |||
| Topical oxybutynin | Anticholinergic.Blocks muscarinic acetylcholine receptor in eccrine sweat glands | May be useful to treat moderate/severe FH | Potential for local irritation, and adverse events such as like xerostomia (rare) |
| Well-tolerated | Limited evidence in children | ||
| Not commercially available in Spain (requires compounding) | |||
| Topical sofpironium | Anticholinergic.Blocks muscarinic acetylcholine receptor in eccrine sweat glands | May be useful to treat moderate/severe AH | Potential for local irritation, and adverse events such as like xerostomia (rare) |
| Well-tolerated | No studies found in children | ||
| Not commercially available in Spain (requires compounding) | |||
| Topical umeclidinium | Anticholinergic.Blocks muscarinic acetylcholine receptor in eccrine sweat glands | May be useful to treat moderate/severe AH and PH | Potential for local irritation, and adverse events such as like xerostomia (rare) |
| Well-tolerated | No studies found in children | ||
| Not commercially available in Spain (requires compounding) | |||
| Oral oxybutynin | Anticholinergic.Blocks muscarinic acetylcholine receptor in eccrine sweat glands | Low cost | Systemic drug |
| Wide availability | Most patients experience adverse events such as xerostomia | ||
| Effective in moderate and severe FH in both childrena and adults | Ill-advised in elderly patients | ||
| Multiple long-term follow-up studies available | Effective only while being administered | ||
| Oral glycopyrrolate | Anticholinergic. Blocks acetylcholine action in eccrine sweat glands | Limited availability in Spain | Systemic drug |
| May be useful to treat moderate and severe FH in children and adults alike | Most patients experience adverse events such as xerostomia | ||
| Ill-advised in elderly patients | |||
| Effective only while being administered | |||
| Botulinum toxin | Temporarily blocks acetylcholine release in sympathetic presynaptic nerve terminals | Effective to treat moderate and severe FH in children and adults alike | High cost |
| Multiple RCTs available | The procedure can be quite painful | ||
| Safe procedure | Requires reapplication a couple of times a year | ||
| FDA-approved to treat AH in adults | Potential serious adverse effects | ||
| Transient muscle weakness | |||
| Iontophoresis | Creates an electric potential gradient that allows the trans-epithelial passage of various solutes, increasing the acidity of eccrine glands, and reducing sweat production | May be effective to treat moderate and severe FH in children and adults alike | High cost |
| It can be synthesized with tap water, aluminum salts, or glycopyrronium | Can be performed at home | Limited availability | |
| Around a dozen devices have been approved by the FDA to treat PH and PPH | The procedure can be painful, or uncomfortable, causing paresthesias | ||
| Potential fo local irritation | |||
| Microwave thermolysis | Destruction of eccrine sweat glands through local heat (cellular thermolysis) | Effective in moderate and severe casesLong-lasting effect (> 12 months)Minimally invasiveThe MiraDry device (Miramar Labs, CA, United States) has been approved by the FDA to treat AH | High costLimited availabilityNo studies in the pediatric populationThe procedure can be painful, cause hyperesthesia, patchy local alopecia, or subcutaneous nodules. Very rare cases of brachial plexus injury with transient neuropathy have been reported. |
| Ultrasounds | Destruction of eccrine sweat glands through focal thermal damage | Long-lasting effect Minimally invasive | High costLimited availabilityNo studies in the pediatric populationThe procedure can be painful, or uncomfortable, cause hematomas, blisters, or hyperpigmentation |
| Radiofrequency | Destruction of eccrine sweat glands through bipolar thermal energy | Long-lasting effect Minimally invasive | High costLimited availabilityNo studies in the pediatric populationThe procedure can be painful, cause erythema or edema |
| Liposuction or curettage | Destruction of eccrine sweat glands. | Long-lasting effect Minimally invasive | High costLimited availabilityNo studies in the pediatric populationThe procedure can be painful, or uncomfortable, cause hematomas, blisters, or hyperpigmentation |
| Sympathectomy, videothoracoscopic sympathectomy | Denervation of eccrine sweat glands. | Effective in moderate and severe forms of FH in both children and adultsMultiple studies available with long-term follow-upsLong-lasting effect | High costLimited availability Anesthetic and surgical risk Postoperative pain may occur, along with hematoma, scars, pneumothorax, hemothorax, subcutaneous emphysema, sensory, and/or limb motor changesHorner's syndrome (mydriasis, ptosis, and anhidrosis)Compensatory sweating and recurrent hyperhidrosis |
AH, axillary hyperhidrosis; FDA, Food and Drug Administration; FH, focal hyperhidrosis; PH, palmar hyperhidrosis; RCT, randomized clinical trials.
Until recently, topical glycopyrronium had to be compounded in Spain, at a high cost, which limited its use. However, in April 2023 it became available in Spain as a pharmaceutical product (Axhidrox, Cantabria Labs), with an annual cost of nearly €200 for the 1st year (and then €160/year). It was already available in, at least, 11 European countries. It has been approved by the European Medicines Agency (EMA) and the Spanish Agency of Medicines and Medical Devices (AEMPS) to treat AH in adults. It is the only topical anticholinergic approved by the AEMPS for the management of AH. Regarding its topical administration, 2 pulses of the product (cream) should be applied to each armpit every night within the first 4 weeks, and then, starting on week 5, 2 pulses per armpit 2 nights/week. Other glycopyrronium formulations can also be commercially found in North America. Thanks to its efficacy and safety profile, we believe it could be considered as a first-line pharmacological treatment for moderate/severe AH in adults (after failed topical antiperspirant therapy). Regarding its use in other parts of the body (palms/soles and craniofacial area), we should mention that most clinical trials and evidence currently available come from patients with AH, and it is not currently approved for use in such locations. However, we believe that the safety profile of topical glycopyrronium, along with logistical issues (cost and availability), would allow its off-label consideration for the management of PH, PPH, and FH with a poor response to other treatments, or in the absence of other therapeutic alternatives. Patients should be advised to avoid touching their eyes with the product, especially when applied to the face or hands, to avoid mydriasis and transient blurred vision. Other therapeutic alternatives that should be considered for different types of FH include oral anticholinergics such as oxybutynin (level of evidence 1- and grade B recommendation), iontophoresis, especially useful in cases of palmar and/or plantar FH, which could be limited by its cost (although not particularly expensive in the mid-term) and availability,24 and BTX-A injections (level of evidence 1+ and grade A recommendation to treat AH), which are highly effective but may also be limited by availability, cost, and (especially in children) the pain associated with the procedure. In refractory cases, various medical devices, and surgical procedures such as sympathectomy can be considered.18
Pediatric FH has been misrepresented in clinical trials despite the impact it can have on this population.24 In this subgroup, the assessment of the risk-benefit ratio plays a predominant role and should be carefully considered.24 (Table 6) Despite the lack of studies in children, antiperspirants with aluminum salts are widely advised as the initial treatment for AH, PH, and PPH in children.24 Topical glycopyrronium has shown good efficacy and an excellent safety profile in 2 RCTs including children,2,34 making it a potential first-line pharmacological treatment for refractory moderate/severe AH in children on topical antiperspirants. Its use has been approved to treat AH, but it can also be a valid alternative to treat PH and PPH. Oral oxybutynin has also been successfully used with good tolerance in children.16 It is advisable to titrate the dose, starting from 2.5mg and up-titrate gradually, while anticipating expected AEs such as xerostomia. Iontophoresis and BTX-A are other alternatives that should be considered. We have not found any trials in children on minimally invasive therapies to be able to make recommendations in this regard. Sympathectomy remains the last resort for non-responders.
LimitationsThis review is limited by its narrative nature and is not a systematic literature review. Additionally, many of the studies analyzed are retrospective in nature, or have heterogeneous methodologies, with various evaluation methods and follow-up periods. Additionally, no well-designed comparative studies have ever been conducted assessing the superiority of one agent over the other. However, we should mention that there is a pretty large number of RCTs available.
ConclusionsFH is a prevalent condition with the potential to impact the patients’ quality of life significantly. Based on safety, efficacy, and cost, we consider that topical glycopyrronium could be the first pharmacological option to treat moderate/severe AH refractory to topical antiperspirants in both children and adults. In other parts of the body, such as palms, soles, or the facial area, topical glycopyrronium could also be considered as an alternative in refractory cases to other treatments, although its use would be off-label. It would be interesting to develop new RCTs to assess its utility in these cases.
FundingNone declared.
Conflicts of interestDaniel Morgado Carrasco, and Raúl de Lucas declared to have received fees from Cantabria Labs.